Antimicrobial Resistance

By Deena Mousa
Executive summary
This is a shallow investigation of antimicrobial resistance. My goal was to estimate the health burden attributable to the condition, learn about barriers to addressing the burden, and identify any promising opportunities that Coefficient Giving could fund. My investigation included a survey of the existing literature and a number of conversations with experts on AMR and related matters.
What is the problem?
Antimicrobial resistance (AMR) occurs when bacteria, viruses, fungi, or parasites evolve to withstand treatments, making infections harder or impossible to treat. This makes routine medical procedures like surgeries or chemotherapy more dangerous and leads to increased mortality, prolonged illness, and higher healthcare costs. In this report, I focus on bacterial infections in particular because bacteria carries the majority of the burden — as I discuss figures around importance and neglectedness going forward, these generally refer to bacterial infections specifically. There are no good estimates for fungal and parasite resistance as far as I am aware. While AMR’s effects are hard to isolate (since it contributes to deaths from many other causes), I estimate that it presents a relevant burden of ~30 million DALYs annually. Based on adjustments to a 2024 Lancet article, which aggregates data sources to model AMR burden, this figure is widely expected to grow. While AMR is a global problem, it disproportionately impacts low- and middle-income countries (LMICs) due to their weaker healthcare infrastructure and higher disease burden. Approximately 90% of the burden of AMR (27 million DALYs) is experienced by people in LMICs.
Who is already working on it?
Governments and philanthropic organizations collectively spend about $1.2 billion annually on AMR, with 65% of this focused on research and development (R&D). Major funders include the Wellcome Trust, Gates Foundation, Novo Nordisk Foundation, NIH, CDC, and BARDA, with most investment directed toward new antibiotics, diagnostics, and surveillance. Public and philanthropic spending on AMR remains low compared to spending on other global health priorities: only $43/DALY overall — comparable with tuberculosis — with an estimated $7–10.50/DALY of that directed toward LMICs.
What could we do?
We could work on interventions related to preventing infections that contribute to the AMR burden, like chlorhexidine for umbilical cord care, typhoid conjugate vaccine scale-up, or infection prevention and control (IPC) bundles in neonatal intensive care units (NICUs). We could also fund work related to improving quality of and access to second-line antibiotics in LMICs, like second-line antibiotic provision for neonatal sepsis in sub-Saharan Africa. There may also be opportunities in surveillance and governance, particularly those directly tied to decision-making, like hospital-level surveillance for resistant bacterial strains. Drug development is another promising approach, especially push and pull mechanisms for narrow-spectrum or neonatal-use antibiotics in late-stage trials, such as supporting a Phase III trial for a narrow-spectrum antibiotic for neonatal sepsis.
Overview
I start with an overview of the importance of antimicrobial resistance and estimate the total importance of AMR at ~$3.1 trillion in “Coefficient Giving dollars” today, with the vast majority coming from health impacts (30 million DALYs, which we value at $CG 100,000 each) and ~$150 billion coming from the direct economic costs of hospitalization. The majority of this burden (27 million CG DALYs, or $2.7 trillion in CG terms) is located in LMICs, with sub-Saharan Africa and South Asia being the hardest-hit regions. This burden is projected to increase substantially over time, though the magnitude and rate of change remain uncertain
I also discuss the current state of funding directed toward antimicrobial resistance, how it is being spent, and the neglectedness of the overall cause area. I estimate public and philanthropic spending related to AMR currently totals ~$1.3 billion annually, with ~$150–350 million of this targeted at LMICs. This equates to ~$43/DALY overall, or $7–10.50/DALY for LMICs. I find that ~65% of public and philanthropic spending on AMR goes toward R&D.
In order to assess the tractability of working on antimicrobial resistance, I begin with a high-level summary to provide a bird’s-eye view of the intervention space. The summary identifies ~50 potential interventions, which I divide into the following categories:
- Prevention: Targeted interventions designed to prevent the infections that contribute most to antimicrobial resistance over time (e.g. sanitation and hygiene, vaccinations)
- Prescribing: Ensuring doctors are able to prescribe antibiotics accurately (e.g. scaling access to diagnostics), and that they prescribe in a calibrated way (e.g. guidance and training to reduce prescription errors, delayed prescribing policies)
- Quality and access: Ensuring that people in LMICs have access to the right antibiotics at the right time and that the drugs are high-quality (e.g. scaling access to existing antibiotics, regulating the quality of antibiotics in LMICs through testing)
- Containment: Limiting environmental transmission of both antibiotics and AMR-resistant strains of infections (e.g. through wastewater, pharmaceutical runoff, or effluent from hospitals and farms)
- Surveillance and governance: Monitoring and understanding the incidence of antibiotic resistance geographically and over time, advocating for optimal regulations and policies around antibiotic use
- Diagnostic and drug development: Directly developing new antibiotics to replace those which bacteria now resist, developing new (e.g. cheaper, faster, more portable) diagnostics to help with more accurate prescribing of antibiotics, implementing mechanisms to fund and encourage antibiotic drug development (e.g. policy, payment mechanisms)
- Animal agriculture: Reducing antibiotic use in animal agriculture settings (e.g. regulating their use to accelerate weight gain, encouraging alternatives), improving animal health to prevent the need for antibiotics (e.g. through increased vaccination or improved sanitation)
I also investigate specific opportunities within each of these areas. Below is a summary table of the preliminary BOTEC results:
I find that prevention and quality and access are the most promising areas for AMR:
- For prevention, the case is strongest where interventions avert infections that are typically treated with antibiotics, and are thus more likely to become resistant — which increases how dangerous an individual case is, and leads to resistance developing faster on the population level. Averting these infections both reduces antibiotic use (limiting the rate at which bacteria evolve) and directly prevents deaths attributable to AMR (since the bacteria being treated with antibiotics are the ones most likely to become resistant to existing and future treatments). In particular, the following stand out as potentially cost-effective prevention opportunities:
- Chlorhexidine (CHX, a disinfectant and antiseptic) for neonatal cord care: CHX is a topical antiseptic applied to the umbilical cord stump of newborns to prevent infection in the days following birth. It directly prevents neonatal sepsis — one of the leading drivers of early-life antibiotic use — while simultaneously reducing mortality.
- Infection prevention and control bundles in neonatal intensive care units: In neonatal intensive care units across LMICs, neonatal sepsis remains a leading cause of mortality. Infection prevention and control bundles — combining low-cost measures like hand hygiene, staff checklists, personal protective equipment (PPE), and decolonization protocols — can prevent the spread of sepsis in these high-risk settings.
- Advocacy for typhoid conjugate vaccine (TCV) scale-up: Typhoid conjugate vaccines (TCVs) are a newer generation of typhoid vaccines that offer longer-lasting immunity and are suitable for infants as young as six months. Pakistan has experienced repeated outbreaks of extensively drug-resistant (XDR) typhoid, making it a priority setting for TCV introduction.
- In the case of quality and access, the core case rests on reducing mortality from resistant infections by ensuring timely access to effective treatment. In settings where first-line antibiotics fail, lack of access to second-line drugs is a direct driver of preventable AMR deaths. Expanding access to proven antibiotics for neonatal sepsis in sub-Saharan Africa, for example, appears to be a cost-effective strategy to reduce the AMR burden.
The next most promising areas within AMR are surveillance and governance and diagnostic and drug development:
- Among surveillance and governance interventions, the impact depends on whether better data leads to faster, more targeted responses. The most compelling interventions are narrow and operationally grounded, such as hospital-based surveillance paired with infection control, which shows strong potential to prevent resistant outbreaks in high-burden settings.
- In diagnostic and drug development, most diagnostics are structurally weak on cost-effectiveness unless tightly tied to AMR-specific DALYs (e.g. diagnostics helping providers treat a condition faster or move to second-line options if the bacterial strain resists first-line treatment). Drug development is more promising, particularly when focused on pushing narrow-spectrum or neonatal-use antibiotics through late-stage trials — this subset of drugs appears to be underfunded globally and is highly relevant to the AMR burden in LMICs.
Based on current evidence, I conclude that prescribing, containment, and animal agriculture are unlikely to be attractive areas for AMR funding, relative to other areas funded by Coefficient Giving.
- When looking at interventions focused on reducing unnecessary prescribing, the health benefits of avoiding unnecessary antibiotics are small and diffuse, with weak links to meaningful AMR burden reduction. Even optimistic BOTECs for diagnostics or nudges in this space fall below our bar for cost-effectiveness. Interventions aiming to improve the accuracy of prescribing by correctly diagnosing resistant strains are likely intractable at the moment — these tests are very expensive, and it appears scientifically infeasible to sufficiently lower those costs.
- In the case of environmental containment, the biological pathways are plausible, but the DALY impact is indirect. Based on current estimates and a sense check, it is unlikely that environmental exposure to antibiotics and resistant strains is a major driver of AMR-related mortality, though evidence is limited. Even under favorable assumptions, interventions like hospital wastewater chlorination do not appear cost-effective.
- Animal agriculture carries many of the same issues as containment, because it involves attempting to reduce the environmental load of resistant bacteria. But on top of this, the tractability is low — especially in LMICs, where food security concerns and weak regulation make policy change difficult. Moreover, the contribution of animal antibiotic use to human AMR disease appears to be modest and indirect, as with prescribing and containment. Antimicrobial resistance’s impact on animal agriculture may place a meaningful economic burden on farmers, though I deprioritize addressing economic impacts. This is partly because I expect economic impacts on animal agriculture to be concentrated among large agricultural firms in high-income countries that already have sufficient profit motive to respond to this.
Ultimately, antimicrobial resistance appears to be a promising cause area — large in scope, neglected relative to its burden, and potentially tractable in key domains. While the evidence base is still developing, the case for targeted funding in prevention and treatment access is strong, and further investigation into promising niches in surveillance and drug development could yield more opportunities.
What is the problem?
Antimicrobial resistance arises when bacteria, viruses, fungi, or parasites evolve such that medications, like antibiotics, become less effective or ineffective. In practice, this is most damaging when bacteria become resistant to key classes of antibiotics.
This can happen through two mechanisms:
- Developing resistance: The use of antimicrobials in humans, animals, and agriculture creates an evolutionary pressure that results in resistant strains of diseases. Overuse and misuse of those antimicrobials accelerates this process.
- Spreading resistance: Poor infection prevention and control (IPC) enables the spread of resistant strains in both healthcare settings (e.g. hospitals) and communities (e.g. person-to-person spread, wastewater).
Antimicrobial resistance renders common infections (e.g. pneumonia, UTIs) harder or impossible to treat and makes essential medical procedures that rely on effective antibiotics (e.g. surgeries, immunosuppression) more dangerous. The economic and social impacts include longer hospital stays, higher treatment costs, productivity losses, and increased mortality.
According to the 2024 Lancet article, only ~26% of global AMR-associated deaths are due to actual resistance — no drug could have addressed the infection. The rest (~74%) are due to a lack of access to effective drugs.
In high-income countries, resistance commonly emerges in hospital settings, where frequent antibiotic use in intensive care units, surgical wards, and long-term care facilities selects for resistant strains. Overprescription, broad-spectrum antibiotic use, and patient demand contribute to the problem, while well-resourced healthcare systems still struggle with ensuring proper antibiotic stewardship. Even with proper antibiotic use, resistance inevitably develops over time. Health care-associated infections (HAIs) affect ~7% of patients in acute care settings in HICs. HAIs are the dominant source of resistant infections in HICs and are a major contributor to AMR mortality in these settings.
In low-income countries, limited access to diagnostics leads to empirical antibiotic use — based on symptoms, rather than confirming the specific infection. Both insufficient access (leading to incomplete treatments) and unregulated overuse (through over-the-counter sales) also contribute to resistance. Widespread availability of counterfeit or substandard antibiotics exacerbates the problem, as less-potent drugs often fail to fully eliminate infections. Poor healthcare infrastructure, overcrowded hospitals, and lack of sanitation enable resistant pathogens to spread more easily. According to the 2024 Lancet article, 7% of patients in acute care settings within HICs have hospital-acquired infections, compared to 15% of patients in LMICs. Resistance rates in LMICs are often higher than in HICs despite lower per capita antibiotic consumption due to weaker infection control, environmental contamination, and poor waste management. However, most AMR-related deaths in low-income countries are still due to lack of access to effective first- and second-line antibiotics, rather than untreatable infections.
In middle-income countries, the story is often a mix of the two above — some patients lack sufficient access to existing antibiotics, while others have infections that resist those antibiotics. Qualitatively, based on interviews with experts, resistance is more common than a lack of access. These settings often experience rising antibiotic consumption as incomes increase, but face gaps in surveillance and stewardship. Moreover, high rates of antibiotic use in animal agriculture (e.g. for prophylaxis and growth promotion) are contributing to rising environmental loads of resistant bacteria and genes.
Agricultural antibiotic use, particularly in livestock for growth promotion, also exacerbates resistance in each of these settings, as resistant bacteria can transfer between animals, humans, and the environment. Projected antibiotic use in livestock — especially in rapidly developing economies — is expected to outpace HIC levels by 2030. Waste from farms and slaughterhouses is a major source of environmental AMR contamination.
The impact of antimicrobial resistance is concentrated — 73% of the health burden is attributable to just six pathogens. The primary drivers of mortality are:
- Klebsiella pneumoniae: Can cause pneumonia, UTIs, and bloodstream infections; spreads primarily through contact with contaminated surfaces or hands.
- Escherichia coli (E. coli): Normally in the intestines; can cause gastroenteritis (food poisoning), UTIs, and occasionally severe systemic infections; typically transmitted through contaminated food or water.
- Staphylococcus aureus (Staph): Causes everything from minor skin infections to severe pneumonia or bloodstream infections. MRSA — which stands for methicillin-resistant S. aureus — doesn’t respond to first-line antibiotics but can often be treated with second-line antibiotics.
- Pseudomonas aeruginosa: Found in soil and water; commonly thrives in moist hospital environments (e.g. sinks, catheters); infects wounds, lungs (especially in cystic fibrosis), and medical devices; known for multi-drug resistance and resilience in harsh conditions.
- Streptococcus pneumoniae (Pneumococcus): Can cause pneumonia, meningitis, and ear infections; spread through respiratory droplets; resistant to penicillin increasingly often.
- Acinetobacter baumannii: Associated with ventilator-associated pneumonia, bloodstream infections, and wound infections in hospitalized patients; frequently multi-drug resistant, requiring last-line antibiotics.
This informs the interventions that are most cost effective, making it more attractive to target individual pathogens than would otherwise be the case.
This section on the different drivers of AMR burden across high-, low-, or middle-income countries heavily references Iskandar et. al (2021), Vikesland et. al (2019), this OECD-WHO briefing paper, and Our World in Data.
What is the health impact of AMR?
The 2024 Lancet article estimates 4.71 million deaths and 178 million IHME DALYs associated with AMR, and 1.14 million deaths and 42.6 million IHME DALYs attributable to AMR in 2021. The latter figures represent a slight decrease from 2019 (5% reduction in attributable deaths, 3% reduction in attributable DALYs).
This equates to 134 million CG DALYs associated with AMR, or 32 million CG DALYs attributable to AMR. I propose a set of conservative adjustments in the ‘Can we trust these figures?’ section below, which updates these estimates to 14 million CG DALYs and 700,000 deaths attributable to AMR, and 66 million CG DALYs and 3 million deaths associated with AMR.
In the ‘Which number should we think of as the burden?’ section, I walk through the calculation to reach my ultimate importance estimate, which is 30 million CG DALYs, or $3 trillion in CG dollars.
What does this mean? The larger number, the deaths and DALYs associated with AMR, compares the observed world against a hypothetical world in which none of the underlying bacterial infections occurred (e.g. no one caught TB). The smaller number, the deaths and DALYs attributable to AMR, compares the observed world against a world with the same incidence of underlying bacterial infection but in which all of the infections are fully drug susceptible (e.g. all TB is drug susceptible).
Which number should we think of as the burden? The right number to consider for our purposes is likely somewhere between the attributable and associated values, but closer to the former. In practice, many AMR-focused interventions (e.g. vaccines, infection prevention) do more than just decrease the number of infections that are resistant rather than susceptible — they can weaken infections or prevent them altogether. As a result, they avert a bit more harm than the “attributable” difference alone — but not as much as the “associated” (eliminating every bacterial infection). The precise ‘correct’ number depends on the type of intervention we are considering (e.g. treating infections addresses a lower total burden than preventing them).
The way to formulate this is:
Total Relevant Burden = Attributable + X*(Associated-Attributable).
The value of X depends on the class of intervention; it will be higher if an intervention is more upstream (e.g. prevents infections rather than ensuring access to second-line treatment). I take 30% as a ballpark figure.
When I take this equation and plug in the reduced estimate for attributable and associated DALYs (explained later on in ‘Can we trust these figures?’), I get:
Total Relevant Burden = 14 million DALYs + 0.30*(66–14 million DALYs) = 14 million DALYs + 16 million DALYs = 30 million DALYs. [All figures are CG DALYs, not IHME.]
How did the burden of AMR change from 2019 to 2021?
In the discussion section, the authors suggest that much of the small drop in AMR mortality from 2019 to 2021 (5% reduction in attributable deaths, 3% reduction in attributable DALYs) is likely connected to COVID-19. In particular, they suggest that widespread infection prevention measures (e.g. PPE, distancing) temporarily reduced the transmission of resistant organisms. Additionally, reduced antibiotic prescribing in high‐income settings, shifts in healthcare utilization, and possible “mortality displacement” (in which some high‐risk individuals died of COVID‐19 instead of bacterial infections) may all have contributed to this short‐term drop. This explanation seems plausible to me, and to the experts I interviewed.
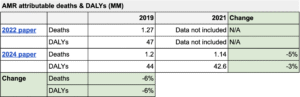
The Lancet paper also provides updated estimates for 2019, which are 6% lower than the previous estimates. That is, they now estimate 1.2 million deaths and 42.6 million DALYs attributable to AMR vs. the previous paper’s estimate of 1.27 million deaths or 47 million DALYs attributable to AMR. This is based on refined modeling, updated antibiotic breakpoints, and the incorporation of newly available surveillance data into their analysis.
How did they get these estimates? The 2024 Lancet article combines mortality data, microbiology datasets, and hospital discharge records to estimate how many sepsis deaths occur around the globe, which syndromes are behind them (e.g. pneumonia, bloodstream infections), and which bacteria cause those syndromes. They begin by building a “sepsis envelope” from national death registries and cause-of-death reports, mapping the fraction of sepsis cases that can be attributed to each syndrome. Next, using tens of millions of lab isolates, they determine which pathogens dominate these syndromes and how commonly each pathogen is resistant or susceptible. Because labs in different countries may measure resistance differently, they adjust the data to a consistent standard for drug susceptibility.
Lastly, to get either the associated or the attributable figure, they apply counterfactuals. If someone would have lived without the bacterial infection, their death is flagged as “associated” with AMR. If someone would have lived as long as the infection was drug susceptible (because they had access to drugs), their death is flagged as “attributable”. By tallying up each infection’s excess mortality and morbidity when it is resistant, they arrive at total deaths and DALYs from AMR in 2021. They then refine the same approach to reevaluate 2019 and compare it with the earlier analysis, updating both antibiotic breakpoints (so that a bug previously classified as “intermediate” might now be “resistant”) and new data sources or countries that were missing from the original analysis.
Can we trust these figures? The authors detail where coverage is thin — particularly in lower-income regions — and use careful modeling to fill gaps, smoothing across neighboring areas and countries. That inevitably introduces uncertainty: incomplete lab data might systematically over- or underestimate resistance, or might not capture community infections as accurately as hospital-acquired ones. The analysis isn’t perfect — no single global data set for AMR is — but it’s likely the best and most transparent snapshot available. The experts I spoke with generally agreed that these figures are the best available and largely reliable (likely not significant over- or underestimates).
Next, a sense check: the Lancet paper estimates 1.14 million deaths attributable to AMR globally on an annual basis. According to the GBD 2019 data, there were ~7 million annual deaths due to bacterial infections. This would imply that the AMR burden is ~15% of the overall bacterial infection burden. Is this realistic? In the chart above, for the microbes that make up most of the AMR burden, the share of deaths from AMR ranged from around ~14% to ~33% depending on the microbe. However, these would represent an upper range, since other bacterial infectious diseases would have lower rates of resistance — and still other infections, from sources like viral illnesses or fungi, would not require antibiotics at all. As a result, I would assume that this estimate is more likely to be too high than too low.
Finally, looking at the modeling decisions. Overall, I believe that this figure is more likely to be an over- than an underestimate. I get to a figure that is closer to 45% of the total, though this is quite uncertain.
Notes on the Lancet model
On counting deaths with multiple causes
For hospital-acquired infections, it often makes sense to attribute the cause of death more to the thing that brought the patient into the hospital, rather than the infection — even if it was the proximal cause of death.
- The Lancet paper uses data on AMR deaths drawn from the Global Burden of Disease (GBD) report, produced by the Institute for Health Metrics and Evaluation (IHME).
- When IHME creates the GBD report, they have to make all the causes of death add up to ~60 million (56.5 million in their 2019 data). As a result, when they integrate the AMR report, they need to “take the deaths” from somewhere else in the GBD.
- From Figure 3, it looks like the answer is mostly going to be lower respiratory infections (a GBD category; 2.5 million deaths, 97 million DALYs), bloodstream infections, and peritoneal and intra-abdominal infections.
- The more that these infections are nosocomial (picked up in hospitals), the more I wonder whether they should be attributed to the cause that brought patients into the hospital (if it was a likely fatal disease/cause), rather than the infection they acquired there (which turned out to be the proximal cause of their deaths).
- This seems particularly clear for diseases like HIV/AIDS, where the immune system is suppressed, and curing the specific infection that hospitalized a patient would not necessarily prevent death from other infections for long.
- I suspect it’s also true for the subset of other hospital patients who end up infected with e.g. MRSA but would have been likely to die either way.
- Another example: if someone gets in a car crash, then gets a blood infection while being treated in hospital, should the cause of death be “Road injury” (a GBD category, with 1.2 million deaths), or MRSA (if that causes the blood infection), or sepsis (if the ultimate cause of death was the patient’s immune system overreacting because of the infection, not the infection itself)?
- GBD would say “Road injury” in this case. I think that is “more right” than saying bloodstream infection, which is “more right” than saying sepsis, because causally preventing the crash will definitely prevent death, preventing MRSA will only potentially prevent death (a different infection might still kill the patient), and preventing sepsis will be even less likely to prevent death (maybe the MRSA still kills them, or they die for another reason).
- Since AMR infections are often picked up in hospitals, and often arise in patients who’ve already received lots of treatment for a disease, the bullet above means the numbers will look inflated if you count every death from someone with an infection as a death caused by infection, rather than sharing “credit” for these deaths between multiple causes (only “counting” as many total causes of death as there are deaths).
- Grounding this, Figure 3 in the 2022 paper shows that lower respiratory infections (LRI) and bloodstream infections (BSI) account for ~57% of AMR-attributable deaths. These are often, but not always, nosocomial. Other syndromes (e.g. TB, UTIs, intra-abdominal infections), which are more often acquired outside the hospital (“community-acquired”), make up the remaining 43%.
- To simplify, let’s assume that 57% of the total DALYs were hospital-acquired and that AMR was decisive in 50% of the cases, and that the 43% were community-acquired and AMR was decisive in 70% of the cases. This 90% figure seems reasonable, given that the individual was unlikely to have been more ill than any other person beforehand.
- To ground the 50% figure: we’d expect this to be lower if many people who die with LRI or BSI in hospitals were already critically ill — with trauma, advanced cancer, AIDS, organ failure, etc. This likely applies more often in high-income country settings.
- However, low-income countries appear to account for much more of the burden, and in those settings much of the AMR burden affects children under five years old. Put another way, there are fewer elderly people with cancer and more neonates with sepsis.
- These figures would imply that 57% of 50% + 43% of 90% = 67% of these cases should qualify as part of the AMR burden. This would suggest a 33% haircut, or a 0.67 multiplier.
Measuring DALY burden for AMR deaths vs. other infections
Relative to deaths from other infections, I think that AMR deaths might have fewer DALYs attached to them than the simple accounting would suggest, since patients being treated for drug-resistant infections are generally in worse physical condition (outside of that particular infection) than people who get drug-susceptible infections.
- This concern focuses not on whether a given death should be attributed to AMR, but on how many descriptive DALYs (years of life lost and lived with disability) should be assigned to each of those deaths.
- My concern is that the Lancet paper overstates the average number of DALYs per AMR-attributable death, by implicitly assuming patients who die from resistant infections would have had long and healthy lives ahead of them — when in fact, many are already severely ill or elderly.
- The Lancet paper’s estimates of 42 million DALYs and 1.14 million deaths implies that each death accounts for 33.6 DALYs, which sounds high given the mortality prospects for many patients with drug-resistant infections. For comparison, typical GBD estimates of DALYs per death vary widely by age and cause: a child who dies in infancy may lose 70–80 DALYs, while an older adult in a hospital might lose fewer than 5.
- To assess whether 33.6 is inflated, it’s useful to look at who’s dying. According to Table 2 of the Lancet paper, just over 21.7% of DALYs attributable to AMR come from children under 5, and the remaining ~78.3% come from individuals aged 5 and older.
- This is important because under-5 deaths account for much higher DALYs per death due to their longer expected life span.
- If most DALYs were coming from under-5s, the average would justifiably be high. But with nearly four-fifths of DALYs coming from older patients — many of whom are in MICs and HICs where deaths often occur in hospital patients with other serious conditions — the average of ~33.6 DALYs per death starts to look too high.
- As a simple check, we could assume that under-5s lose ~65 DALYs per death and that the 5+ group loses ~25 DALYs per death on average (reflecting comorbidities, hospital settings, and older age). Weighted by the 21.7%/78.3% age split, that gives a composite DALY-per-death of (0.217 × 65) + (0.783 × 25) = ~33.6 DALYs per death — this is the paper’s estimate.
- We might think that the 5+ group averages less than 25 DALYs per death, given that many resistant infections occur in older adults or hospitalized patients with poor baseline health. If we instead assume this group averages 20 DALYs per death, the weighted average drops to (0.217 × 65) + (0.783 × 20) = ~26.7 DALYs per death. That would imply a 21% haircut, or a multiplier of 0.79. If we use 15 DALYs per death for the 5+ group, the average drops to ~22.2, implying a 34% haircut, or multiplier of 0.66.
- I would suggest a ~25% haircut (0.75 multiplier), depending on your assumptions about the typical adult dying from AMR.
Measuring the benefit of making resistant cases susceptible to drugs
The model may use an overly generous estimate of the benefit of turning resistant cases into susceptible cases because it may be over-anchoring to the magnitude of that benefit in high-income countries.
- The model generates its main counterfactual as “what would happen if you replace these drug-resistant cases with drug-susceptible cases, for each infection?”
- In high-income countries (HICs), people with drug-susceptible infections are almost always treated, and rarely die. This makes it very valuable to change drug-resistant infections into drug-susceptible ones. In low-income countries (LICs), treatment isn’t as reliably available, so it matters less whether an infection is drug-susceptible.
- Though note there are factors going the other way. For example, it is cheaper to treat drug-susceptible TB than drug-resistant TB, and that difference matters more in LICs — in that sense, an infection being drug-susceptible might have a bigger impact in an LIC vs. an HIC.
- Data from HICs tends to be higher-quality, so it’s likely that HIC data is overrepresented in the model — and thus, that the model overestimates the benefit of making an infection drug-susceptible.
- That said, this model doesn’t apply a single relative risk for resistance across geographies. The Lancet article explains that its model estimates infection-specific and region-specific case fatality rates (CFRs) for both resistant and susceptible cases, and that they were informed by both published studies and hospital-based data.
- That said, the underlying concern still stands: because data quality is significantly better in HICs, the best-informed estimates of the susceptible/resistant CFR gap are disproportionately based on HIC settings.
- These high-quality HIC data may, in turn, anchor the RR estimates, even when applied to regions where both susceptible and resistant infections have high fatality rates. In other words, even if the model varies its inputs by region, the relative difference between CFRs may still be overstated in LMICs because the most reliable data informing the model come from richer countries.
- Based on the table in ‘How much of this is attributable to the burden in LMICs?’, sub-Saharan Africa and South Asia together make up ~50% of the burden. One might assume that the benefit of turning resistant cases susceptible has been overestimated for these regions by ~20% and take a haircut of 20% of 50% = 10%. As a result, I suggest a multiplier of 0.9.
Here is a table of my estimates for the ultimate attributable importance figure in millions of deaths and CG DALYs:
![]()
I also make the appropriate adjustment for the associated burden (excluding the third haircut, because it only applies to the attributable burden):
![]()
Note, however, that the pure ‘attributable’ burden is an underestimate of the relevant health burden. It assumes that interventions simply convert infections from resistant to susceptible — but many of the interventions we consider are further upstream, preventing infections entirely. As a result, as noted above, I take these attributable estimates and adjust upward to account for the burden that could be addressed via prevention.
We extracted appendix tables from the 2022 Lancet table, including a more detailed regional breakdown of attributable mortality in. Based on this data, ~11% of the AMR-attributable burden is in HICs and ~89% is in LMICs. Given the 2024 paper’s estimate of 32 million CG DALYs or $3.2 trillion in CG importance attributable to AMR, this would imply 28.5 million DALYs or $2.85 trillion in CG importance attributable to AMR in LMICs. (Other than the “high-income” entry below, all data represents LMICs.)
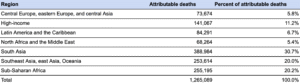
The paper shows that sub‐Saharan Africa and South Asia together account for the highest per‐capita AMR burden, while southeast Asia and parts of Latin America also account for large shares of the total mortality.
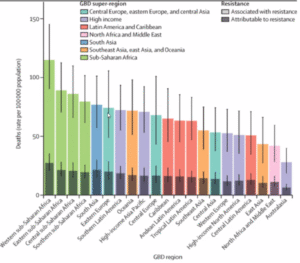
Within Africa, the west and central regions (e.g. Nigeria, DR Congo) stand out for having both high infection rates and limited access to quality antibiotics. Meanwhile, India consistently shows up as a hotspot in South Asia, driven by high antibiotic consumption and widespread hospital‐acquired pathogens. Middle‐income regions like parts of Latin America (especially tropical Latin America) also see considerable levels of drug‐resistant infection, but typically not at the per‐capita extremes of sub‐Saharan Africa or South Asia.
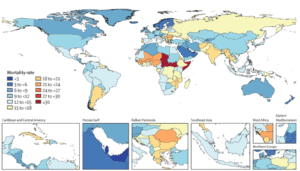
The Lancet forecasts estimate that global AMR‐attributable mortality will rise from around 1.14 million in 2021 to nearly 1.9 million by 2050. Although the paper does not explicitly separate LMIC vs. HIC trends, their text and figures suggest that LMICs share of the burden will grow faster, owing to rising antibiotic use (often without robust stewardship), more hospital crowding, and these countries’ rising share of the global population. South Asia, for instance, is predicted to have some of the steepest increases in AMR‐related deaths, and sub‐Saharan Africa remains at very high per‐capita risk. As a result, the lion’s share of the future incremental rise seems likely to accrue in LMIC countries, pushing the AMR death toll there to well over a million annually by 2050.
What is the economic impact of AMR?
A Center for Global Development (CGD) report published in September 2024 estimates that antimicrobial resistance currently costs about $66 billion per year in extra inpatient expenses globally (or 0.7% of annual health spending).
- If no policy changes occur (the “business as usual” scenario), CGD estimates that the direct cost of AMR could rise to $159 billion by 2050, whereas an “accelerated” rise in resistance could push costs to $325 billion.
- On top of these direct health costs, AMR diminishes economic output by shrinking labor participation, potentially leading to future global GDP losses of $1.7 trillion in the accelerated scenario.
- However, for this investigation, I’ll set aside future projections and focus on integrating present-day estimates into our estimate of AMR’s burden.
As explained below, adding direct economic costs (i.e. healthcare costs) to health effects would roughly bring the total attributable burden of AMR in CG terms from ~$3.2–$3.34 trillion.
See the appendix for a discussion of the CGD report’s methodology, and the degree to which I think their estimates are reasonable.
How much are health and economic impacts likely to increase over time?
The authors of the Lancet paper project that by 2050, approximately 1.9 million deaths per year will be directly attributable to drug-resistant bacterial infections (versus ~1.14 million in 2021), and 8.2 million will be associated with resistance. This represents a 66% increase in mortality burden over 29 years, or a 1.8% compound annual growth rate (CAGR).
How did they get this figure? To project AMR mortality through 2050, the authors used the GBD forecasting framework, which first projects future deaths by syndrome (e.g. pneumonia, bloodstream infections) and pathogen (e.g. E. coli) using time-series models trained on historical trends. They then modeled future resistance rates by pathogen-drug pair, drawing on past resistance data and covariates like antibiotic consumption, healthcare access, and GDP per capita. They overlaid these projected resistance rates onto the mortality forecasts to estimate how many future deaths would involve resistant infections. Finally, they applied previously estimated excess mortality risks for resistant vs. susceptible infections to compute the share of deaths directly attributable to resistance, across five scenarios ranging from optimistic (e.g. better access to second-line antibiotics) to pessimistic (e.g. accelerated resistance growth).
Can we trust the methodology? Overall, the authors’ process is more data-rich than those used to create older global AMR forecasts, drawing on hundreds of millions of microbiology records to tune the baseline. They also apply spatiotemporal smoothing to fill gaps for countries lacking direct surveillance, which is a reasonable approach but inevitably injects uncertainty — especially in low- and middle-income regions with poor lab data. They may overestimate or underestimate if the real-world distribution of resistant pathogens differs significantly from what the smoothing models imply. Their forward-looking estimates depend on GBD cause-of-death forecasts that themselves carry uncertainty about how economies, demography, and health systems will evolve. Still, this is the most comprehensive public model to date, so while the confidence intervals are wide, there is no obvious reason to adjust the estimate in either direction.
Is the estimate reasonable? The Lancet paper also estimates that 1.06 million deaths were attributable to AMR in 1990. If the 1990 and 2021 figures are both accurate, this would represent a 7.5% increase over 31 years — compared to an estimated 66% increase over the next 30 years. This makes me instinctively a bit skeptical of the projection. One would expect the rate of change to increase over time due to an aging world population that is at higher risk from infections, and rising antibiotic use (especially in regions with weaker stewardship). However, this jump seems a bit too high outside-in.
Overview
I estimate that public and philanthropic spending related to AMR currently totals ~$1.3 billion annually, with ~$200–300 million of this targeted at LMICs. I very roughly estimate an additional ~$1.3 billion in related private sector spending. Below I break down where philanthropic, government, and private sector funding comes from and where public and social sector funding is going.
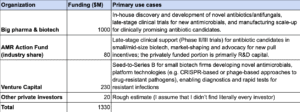
- Philanthropic funding: ~$150 million in annual funding, primarily on R&D “push” incentives (e.g. preclinical or early clinical research) and policy or capacity-building initiatives.
- Government funding: ~$1.2 billion in annual funding, largely to basic research and early-to-late-stage product development. Most of this goes to research institutions and biotech companies focused on novel antibiotics, diagnostics, and vaccines. I’d estimate that only roughly 10–15% of this is going to LMICs.
- Private sector funding: ~$1.3 billion in annual funding, mostly to R&D and clinical trials for novel antibiotics and antifungals, aiming to address market gaps in high-income countries.
Data from the Global AMR R&D Hub shows that in 2020 — the most recent year with available data — $1.5 billion was invested in antimicrobial resistance R&D. This figure is relatively consistent with historical (pre-COVID) reports, e.g. the 2018 report. This implies that ~65% of annual spending on AMR (~$2.3 billion) targets R&D, and the other ~35% (~$0.8 billion) is spent on other interventions, including those related to healthcare systems.
Note that these figures are approximations; several of the single-year philanthropic estimates were derived using an institution’s biggest public multi-year commitments, rather than by directly using annual budgets. The private sector figures are especially low-confidence, but less central to neglectedness — since private firms are less interested in global public health than other philanthropic funders, their funding is less likely to cover the same things we’d want to support.
How does this compare to other cause areas?
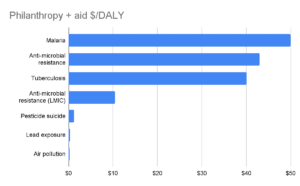
Based on an annual burden of 32 million DALYs, the ratio of philanthropic and public spending to health burden is ~$43/DALY. The ratio is ~$210/DALY for HIV/AIDS, ~$50/DALY for malaria, and $40/DALY for tuberculosis.
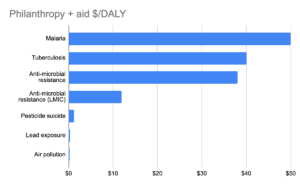
Based on the estimated burden (28.5 million DALYs) and funding amount ($200–300 million) for LMICs, this would provide an approximate range of $7–$10.50/DALY, making AMR in LMICs likely more neglected than in HICs on the basis of public and philanthropic funding. This would also make it more neglected than many of the areas Coefficient Giving currently funds, which suggests that there may be cost-effective opportunities in the space.
The LMIC-specific estimate is based on assuming that approximately ~$110–150 million (10–15%) of global government funding for AMR, along with ~$100–150 million of the $150 million in philanthropic funding, is targeting LMICs. See here for more detail.
Where does this funding go?
The majority of these funds (I’d estimate roughly 65%) are devoted to research and development (R&D) of new antibiotics, diagnostics, and other tools, while much smaller shares support on-the-ground capacity building, stewardship programs, policy advocacy, and ensuring access to solutions. Funding is mostly driven by challenges in high-income countries, with a small portion targeting low- and middle-income countries.
Caveat: AMR-adjacent spending
Note that there is significant additional funding that is not targeted directly at AMR, but which reduces the burden of AMR. For example, water, sanitation and hygiene (WASH) interventions reduce the incidence of disease and therefore slow the development of AMR. One estimate of funding in WASH placed it at ~$1.4 billion. When we exclude funding for these AMR-adjacent areas, we overestimate how neglected AMR actually is.
Philanthropic funding
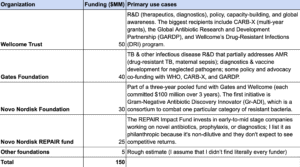
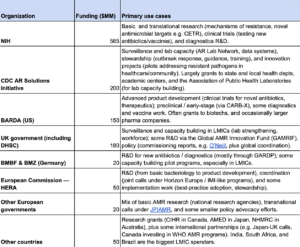
I estimate that approximately ~$90 million in annual philanthropic funding targets antimicrobial resistance. The largest funders in this space are the Wellcome Trust (~$40 million) and the Novo Nordisk Foundation (~$25 million). The majority of this funding is spent on R&D “push” incentives (e.g. preclinical or early clinical research) and policy or capacity-building initiatives.
See here for references.
Government funding
Note: This section was written before a series of steep funding cuts for the NIH and CDC. I still believe the NIH and CDC to be among the largest funders of AMR work, but specific figures and examples may be outdated.
I estimate that government funders spend approximately ~$1.2 billion each year to target antimicrobial resistance. The largest funders in this space are the NIH, CDC, and BARDA. Most of this funding goes to support research institutions and biotech companies conducting basic research and product development in high-income countries
I’d estimate that roughly 10–15% of this (~$110–150 million) is going to LMICs. While US government agencies like NIH, CDC, and BARDA are major funders of AMR work, the bulk of their funding is directed toward research institutions, surveillance infrastructure, and biotech companies within the United States. These investments are often focused on:
- Basic research into resistance mechanisms
- Early-to-late stage product development (e.g. antibiotics, diagnostics, vaccines)
- National surveillance and infection control programs
- Strengthening domestic pandemic preparedness and biodefense (e.g. BARDA’s mission)
Their default orientation is toward protecting the health of Americans and making it easier for US companies to innovate, rather than reducing global burden.
However, a nontrivial minority of AMR-related funding flows to LMICs. For example:
- NIH supports some collaborative research and capacity building in LMIC settings, particularly around TB, malaria, and hospital-acquired infections.
- CDC funds global health programs, including surveillance networks (e.g. the Global Antimicrobial Resistance Surveillance System, GLASS) and technical assistance for LMIC governments.
- BARDA’s work is primarily focused on the US, though it supports product development with global implications
This 10–15% range reflects both intentional LMIC-targeted programs and benefits from HIC-focused work that spills over to LMICs, but still recognizes that US government AMR funding remains overwhelmingly focused on HICs (e.g. the United States).
See here for references.
How much is this likely to be reduced by aid cuts?
At present, HIC government funding for many forms of medical research seems likely to be reduced over the next few years. Unfortunately, this factor is highly uncertain. The US government has signaled it will continue funding global health programs, but the scale of its support remains unclear pending Congressional appropriations bills, court cases that will shed light on the extent to which the executive can unilaterally cancel funding, and future rescissions packages that may be sent to Congress.
Where does public and philanthropic funding go?
A few key organizations together receive a significant portion of public and philanthropic funding:
- Combating Antibiotic-Resistant Bacteria-Xcelerator (CARB-X): CARB-X has received a total of $453 million (2016–2024) in funding and invests mostly in early-stage therapeutics, as well as new vaccines and diagnostic tools that are prioritized for responding to antibacterial resistance.
- Historically, their funding has come from BARDA (US government), the NIH (in-kind contributions), the Wellcome Trust, the Bill and Melinda Gates Foundation, and official development assistance (ODA) from Germany and the UK.
- Most funding is provided to private companies, particularly small biotech firms. In exchange for the funding, CARB-X requires companies to make their products accessible and affordable to people in LMICs.
- Global Antibiotic Research and Development Partnership (GARDP): GARDP receives ~$30–50 million annually in funding, and focuses on developing new antibiotic treatments. GARDP has dedicated programs for sexually transmitted infections, sepsis in newborns and children, and serious bacterial infections in adults.
- The Fleming Fund: A UK aid program that distributes ~$50 million annually, with a focus on AMR surveillance in LMICs funded by the Department of Health and Social Care (DHSC).
- The Fleming Fund’s initial funding phase, totaling £265M, ran over a five-year period (2016–2021) but was extended to March 2023.
- Phase 2 is funded for £210 million over 2022–2025. The Fund’s goal is to support LMICs in generating, sharing, and using antimicrobial resistance data.
- The AMR Action Fund: ~$125 million in annual funding from a combination of philanthropic, multilateral, and private sector funding, invested in companies developing new therapeutics.
Private sector investment
I estimate that private organizations spend (very) approximately ~$1.3 billion each year to target antimicrobial resistance, mostly through R&D and clinical trials for novel antibiotics and antifungals, aiming to address market gaps in high-income countries.
See here for references.
Overview
Based on interviews, desk research, an intervention summary, and initial BOTECs, I’ve identified prevention and quality and access as the most promising areas for AMR interventions.
Prevention efforts like neonatal antiseptics or vaccines often reduce antibiotic use while averting deaths from infections where resistance is common or access to treatment is limited. Many are already well-evidenced and scalable, making them tractable even in low-resource settings.
Quality and access interventions — such as ensuring the availability of effective second-line antibiotics — can directly avert deaths from drug-resistant infections.
Unlike more diffuse strategies, these both target the health burden of AMR head-on, and BOTECs suggest they may exceed Coefficient’s cost-effectiveness bar.
The next most promising areas are surveillance and governance and diagnostic and drug development. These are more heterogeneous: some interventions, like infection surveillance tied to outbreak control or trials for narrow-spectrum neonatal antibiotics, show high potential, while others (e.g. national dashboards or broad diagnostic rollouts) are unlikely to meet cost-effectiveness thresholds unless they are designed with a strong link to policy or behavior change.
In contrast, prescribing, containment, and animal agriculture are unlikely to be cost-effective or tractable avenues to alleviating the burden of AMR. Prescribing-focused interventions often reduce antibiotic use without a clear link to health gains; containment suffers from long causal chains and high costs; animal antibiotic use — while high in volume — appears to contribute only marginally to avertable human disease burden, with limited levers for cost-effective change in LMIC settings.
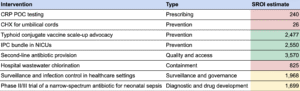
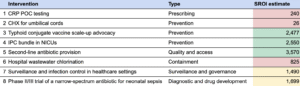
The table below shows a set of interventions for which I created BOTECs, because they seemed like the most promising of each category among the ones I found:
In each of the sections below, I:
- Lay out the overall theory of change and scope for this category of intervention, including a high-level summary of potential interventions.
- Identify and explore the crux(es) that determine whether this area seems sufficiently cost-effective to be worth funding, and form an overall view on the area’s attractiveness.
- Provide BOTEC(s) on specific interventions within the category, to understand the value a grant might provide, selecting for grants I felt were indicative of the particular value and challenges of the category.
See here for a complete list of interventions I found. In general, I’ve identified interventions in this section through a combination of desk research and interviews and explored the ones that had a strong evidence base and/or seemed like they could potentially deliver a high SROI.
Prevention
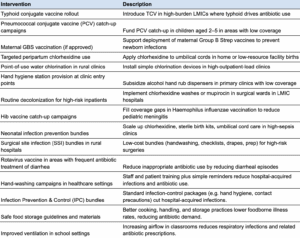
Prevention opportunities are targeted interventions designed to prevent the infections that contribute to the burden of antimicrobial resistance by causing deaths and driving increased resistance over time. Many prevention interventions — such as vaccines, WASH, and topical antiseptics — have clear and well-documented health benefits. The key uncertainty is whether the proportion of their value that addresses the AMR burden is large enough to justify these interventions on AMR grounds. If so, that could push us to see AMR as a more promising area; if not, then the strength of these interventions shouldn’t update us one way or the other.
There are two main ways prevention may reduce the AMR burden:
- Reducing the transmission of resistant pathogens, which prevents infections that are typically treated with antibiotics (e.g. neonatal sepsis, respiratory infections), thus limiting the burden of AMR disease.
- Reducing direct deaths that are attributable to AMR, for example by preventing an infection that would later drive mortality due to (a) being resistant to existing antibiotics, or (b) the individual not having access to antibiotics that work.
AMR work is often framed as driven by the desire to slow the emergence of resistance. However, much of the health burden of AMR comes from infections that are already resistant, or the lack of access to high-quality antibiotics for infections that aren’t resistant. In both cases, addressing the problem can be highly cost-effective even if it doesn’t do much to change the underlying rate of resistance.
Prevention is a viable and, in some cases, highly attractive category of AMR interventions — particularly when it targets pathogens where resistance is a major driver of mortality. Interventions like typhoid conjugate vaccines and chlorhexidine for neonatal cord care directly avert infections that are often treated with antibiotics, and in doing so, they reduce both antibiotic use and the burden of drug-resistant disease. In high-resistance contexts, a substantial share of the deaths averted may be attributable to AMR: for example, typhoid in Pakistan is ~80% extensively drug-resistant (XDR), and neonatal sepsis pathogens like Klebsiella and E. coli are among the leading global causes of AMR-attributable death. Even when infections are caused by drug-susceptible strains, lack of access to appropriate antibiotics in many LMICs often leads to the same kinds of outcomes seen with resistant strains. This can blur the line between AMR and non-AMR deaths.
While most prevention interventions would be above the bar on health grounds alone, a meaningful portion of their impact — often 20–40% in the best cases — may reasonably be considered as addressing AMR burden (think of this as the difference in impact between not getting a disease and getting a disease that is then treated with working antibiotics). This makes prevention a strong candidate for inclusion in an AMR-focused portfolio.
Below is a selection of potential interventions, and an exploration of two prevention interventions — chlorhexidine (CHX) for neonatal cord care and advocacy for typhoid conjugate vaccine scale-up — to illustrate how this tradeoff (between counting and not counting impact as AMR-related) plays out in practice.
Intervention: Chlorhexidine for umbilical cords
Chlorhexidine (CHX) is a topical antiseptic applied to the umbilical cord stump of newborns to prevent infection in the days following birth. In low-resource settings, particularly where births occur at home or in facilities with limited sanitation, umbilical cord infections are a major contributor to neonatal mortality. CHX is simple to apply, requires minimal training, and has been shown in multiple randomized trials to significantly reduce both infections and neonatal deaths. CHX is part of WHO guidelines and has widespread policy adoption, but there are settings where delivery remains constrained.
CHX stands out as a promising targeted prevention intervention for AMR. It directly prevents neonatal sepsis — one of the leading drivers of early-life antibiotic use — while simultaneously reducing mortality. Based on a BOTEC using trial-based effect sizes and current cost estimates, CHX for neonatal cord care comfortably clears Coefficient’s 2100x bar on mortality reduction alone. Although its second-order AMR benefits are modest by comparison, they reinforce the case for this intervention as an unusually cost-effective opportunity.
Based on a BOTEC, CHX treatment to umbilical cords for neonatal care may have an SROI of 4,131x. I estimate that ~25% of this impact is attributable to reducing AMR-driven health burdens. See here for the full BOTEC.
I modeled a potential intervention to scale chlorhexidine (CHX) application for neonatal cord care in low-income countries, targeting settings with high neonatal mortality and limited hygiene during home births.
- I assumed the intervention would reach 1 million newborns annually, consistent with a plausible rollout size for a national or regional program.
- I used the UN Inter-agency Group for Child Mortality Estimation’s figure of 2.7% neonatal mortality as a baseline for high-burden settings.
- That implies 27,000 expected neonatal deaths in the target population absent the intervention.
- I drew on pooled effect estimates from Arifeen et al. (2012), Mullany et al. (2006), Soofi et al. (2012), Sazawal et al. (2016), and Semrau et al. (2016), which suggest that CHX cord care reduces neonatal mortality by approximately 15%
- Applying that reduction yields ~4,050 neonatal deaths averted. I used an estimate of 51 DALYs per neonatal death to convert this to 206,550 DALYs averted. At $100,000 per DALY, this corresponds to ~$20.7 billion in Coefficient value.
- I used GiveWell’s estimate of $5 per CHX treatment (which includes both the direct cost of the treatment and the implementation cost), for a total cost of $5 million per year
- That yields an SROI of 4,131x.
I also considered the second-order AMR-related benefits of CHX by modeling its impact on downstream antibiotic use:
- Assuming 2.8% of newborns develop infections (28,000 cases per million births), and 50% of those cases are treated with antibiotics based on Berhane et al., (2021) and Applegate et al., (2020), that implies 14,000 baseline antibiotic courses.
- I modeled a 23% reduction in infections due to CHX, based on pooled RCT findings, which yields 6,440 infections (and thus 3,220 antibiotic courses) averted.
- Using an estimate of 0.01 DALYs averted per antibiotic course avoided (sourcing and discussion in the prescribing section), this adds ~32.2 DALYs.
- This corresponds to $3.22 million in CG value — tiny relative to the mortality gains.
- This component alone yields a very low preliminary SROI (~0.64x) and would not be cost-effective if pursued solely for its AMR impact.
How much of this is relevant to the AMR burden?
- CHX reduces deaths from neonatal sepsis, especially due to pathogens like Klebsiella pneumoniae, Staph aureus, and E. coli.
- These pathogens are among the top contributors to neonatal deaths caused by AMR.
- The 2022 Lancet study found:
- K. pneumoniae caused 80,000+ neonatal deaths globally attributable to AMR.
- The burden is highest in South Asia and sub-Saharan Africa — the same regions where CHX is most used.
- I assume that 20–30% of neonatal sepsis deaths in high-AMR settings are caused by drug-resistant infections.
- Since CHX reduces infection risk regardless of drug resistance, it prevents both susceptible and resistant infections — but we care most about the infections that would have been fatal due to AMR.
- If CHX averts 4,050 neonatal deaths per year, and ~25% of those would’ve been due to resistant pathogens, then ~1,000 deaths averted are AMR deaths.
Who could we fund to do this? PATH does significant work around advocacy and implementation for the use of CHX in neonatal care in sub-Saharan Africa.
Intervention: Typhoid conjugate vaccine scale-up advocacy in Pakistan
Typhoid conjugate vaccines (TCVs) are a newer generation of typhoid vaccines that offer longer-lasting immunity and are suitable for infants as young as six months — a major advantage over previous typhoid vaccines. They are safe, effective (~80% efficacy), and recommended by the WHO for use in endemic countries. Typhoid itself is a waterborne bacterial infection caused by Salmonella enterica serovar Typhi, which poses a serious public health threat in low-income countries with inadequate sanitation infrastructure.
Pakistan has experienced repeated outbreaks of extensively drug-resistant (XDR) typhoid, making it a priority setting for TCV introduction. In this BOTEC, I’m not modeling the cost of delivering TCVs directly, but rather estimating the value of funding policy advocacy that successfully accelerates nationwide TCV scale-up — for example, via a nonprofit that pushes for government adoption and rollout.
This is a useful lens for AMR-related work because (a) typhoid is a disease with known and growing antibiotic resistance concerns, and (b) policy wins can be extremely cost-effective if they catalyze large-scale public delivery of proven interventions.
I estimate that advocacy for a scale-up of typhoid conjugate vaccines in Pakistan may have an SROI of ~2,477x. I estimate that ~25–40% of this is addressing the health burden of AMR. See here for the full BOTEC.
I modeled a scenario where Coefficient funds policy advocacy in Pakistan that successfully accelerates the scale-up of TCVs nationwide.
- Pakistan’s total population is 225 million and the annual incidence of typhoid is ~0.49% (based on regional studies), resulting in ~1.1 million cases per year.
- Using a case fatality rate (CFR) of 0.56%, this implies ~6,174 typhoid deaths annually.
- TCV is approximately 80% effective, and I assume a successful government scale-up would achieve 80% coverage, based on precedent and WHO goals.
- That implies 705,600 cases averted and ~3,951 deaths averted per year.
- Using an estimate of 40 DALYs per death, this implies 158,054 DALYs averted.
- At Coefficient’s $100,000 per DALY valuation, this equates to ~$15.8 billion in CG value.
- I assume the advocacy campaign would cost $2 million to fund and would speed up the rollout by 3 years, and that Coefficient would receive 10% causal credit for the change.
- Applying both adjustments (speed-up and credit attribution), the adjusted CG impact is ~$474 million.
- Dividing by the $2 million cost yields an SROI of ~2,371x.
However, note that the value of this intervention depends significantly on the counterfactual use of the funds. For example, if the government would otherwise spend this money on something appreciably lower in SROI, this intervention would look better given the sizable difference in expected impact. If the money would instead be used on other interventions with higher cost-effectiveness, closer in value to TCV, the grant would look significantly worse.
I also modeled the benefits of avoiding a course of antibiotics and therefore slowing the development of AMR.
- I started with the same number of antibiotic courses averted as typhoid cases averted from the primary BOTEC: 705,600. This assumes each typhoid case typically results in one course of antibiotics, usually fluoroquinolones or cephalosporins.
- I estimated that each antibiotic course averted would avert 0.01 DALYs (sourcing and discussion in the prescribing section). This is a mid-range, slightly optimistic estimate drawn from the best available literature (e.g. Dickinson et al. (2021)., Atkins et al. (2021, UK)).
- Multiplying these together gives 7,056 DALYs averted annually from reduced antibiotic use due to the vaccine.
- I assumed a 3-year speed-up in implementation due to successful advocacy, so I multiplied the annual benefit by 3 to get the total value.
- I also assumed 10% attribution to the advocacy effort, meaning Coefficient would only take credit for 10% of the value generated.
- At a value of $100,000 per DALY, this yields a Coefficient impact of ~$211.7 million for the AMR benefits.
- Given the $2 million cost of the advocacy campaign, this gives an AMR-only SROI of ~106x — a meaningful supplement to the health-based case, though not nearly enough on its own to clear the Coefficient bar (~2100x).
How much of this is relevant to the AMR burden?
- Pakistan has been the epicenter of extensively drug-resistant (XDR) typhoid, especially in Sindh province.
- According to the CDC and Gavi, by 2020 over 80% of typhoid isolates in Karachi were XDR — resistant to fluoroquinolones and third-generation cephalosporins.
- In those cases, first-line treatments fail, increasing both case fatality rates and complications.
- TCV reduces overall typhoid incidence — and in doing so, prevents infections that would have been difficult or impossible to treat with antibiotics.
- If 80% of typhoid cases are XDR, and you avert 3,951 deaths total, roughly 3,000 deaths averted are resistant-strain deaths.
- This could suggest that 75–80% of DALYs would be AMR-related, but that may overstate things:
- Some patients would have accessed carbapenems or other advanced treatments that work against XDR cases.
- Some deaths might still occur even with drug-susceptible strains due to late presentation, poor access, etc.
- A conservative but reasonable estimate is that ~40% of the SROI should be attributed to reducing AMR health burden, given very high resistance prevalence, direct impact on a high-burden resistant disease, and the prevention of deaths that would have otherwise been unavoidable.
Who could we fund to do this? Sabin Vaccine Institute — which has experience with vaccine policy advocacy, including typhoid — or Gavi CSO Constituency.
Intervention: infection prevention and control bundle in neonatal intensive care units
In neonatal intensive care units across low- and middle-income countries (LMICs), neonatal sepsis remains a leading cause of mortality. The risk of infection is elevated due to fragile immune systems, frequent medical interventions, and suboptimal hygiene practices. Infection prevention and control bundles — combining low-cost measures like hand hygiene, staff checklists, personal protective equipment (PPE), and decolonization protocols — offer a pragmatic, facility-level solution to avoid the spread of sepsis in these high-risk settings. They have been shown to significantly reduce sepsis incidence in LMIC hospitals.
I modeled a potential intervention to implement infection prevention and control bundles in neonatal intensive care units across LMICs, finding that it may have an SROI of 2550x. See here for the full BOTEC.
- I assume the intervention would reach 1 million neonatal intensive care unit admissions annually, across high-burden regions.
- I use a baseline neonatal sepsis rate of 10%, implying 200,000 sepsis cases per year among that group, referencing Zaidi et al. (2005)’s 20% estimate and discounting by 50% to account for uncertainty around the figure and to be conservative.
- The baseline mortality rate for neonatal sepsis cases is 30% based on Zaidi et al. (2005), implying a counterfactual of 60,000 deaths.
- I assume that the infection prevention and control bundle would reduce neonatal sepsis mortality by 25% relative to baseline, leading to a mortality rate of 22.5% and 45,000 deaths.
- Jozwiak, et. al (2016) review the literature on the implementation of sepsis care bundles, summarizing observational studies and meta-analyses that linked bundle adherence to improved patient outcomes. They cite a systematic review of 50 studies showing that implementation of sepsis bundles was associated with a 34% reduction in odds of death (OR 0.66; 95% CI: 0.61–0.72), and emphasize that higher bundle compliance — especially early and complete adherence — is associated with lower mortality. They reference multicenter data from Levy et al. (2015), which found that moving from low- to high-compliance ICUs was associated with a 25% relative reduction in sepsis mortality.
- Cardoso, et. al (2010) conducted a prospective, multicenter cohort study of 897 ICU patients with community-acquired severe sepsis or septic shock in 17 Portuguese ICUs. They tracked adherence to a “core” 6-hour sepsis bundle (lactate measurement, blood cultures, early antibiotics, fluids, vasopressors, etc.) and found that full compliance with the bundle was associated with a 56% reduction in 28-day mortality (adjusted OR 0.44; 95% CI: 0.24–0.80), equivalent to a number needed to treat (NNT) of 6.
- Averting 15,000 infant deaths is associated with 765K DALYs, or a CG value of $76.5 billion.
- I assumed a cost of $15 per infant for the infection prevention and control bundle, including consumables and staff training, leading to a total cost of $15 million. The $15 per infant estimate is consistent with programmatic data from WHO-endorsed infection prevention and control bundles (Dramowski et al. (2022)) and a 2021 Indian neonatal intensive care unit initiative, which report costs ranging from $12–$15 per infant for training, consumables, and hygiene infrastructure.
- This gives an overall SROI of 2550x.
How much of this is relevant to the AMR burden?
- Infection prevention and control bundles reduce neonatal sepsis from all causes — but in LMIC neonatal intensive care units, a large share of sepsis is caused by drug-resistant pathogens.
- Zaidi et al. (2005) report that pathogens like Klebsiella pneumoniae, E. coli, Acinetobacter spp., and Staphylococcus aureus dominate neonatal bloodstream infections in LMIC hospitals.
- GRAM 2022 estimates that K. pneumoniae alone caused 80,000+ neonatal deaths globally attributable to AMR in 2019 — with the burden concentrated in South Asia and sub-Saharan Africa.
- Resistance to first-line antibiotics (ampicillin + gentamicin) is common in neonatal intensive care units: Zaidi et al. (2005) estimate that up to 70% of neonatal pathogens in LMICs are not covered by this regimen.
- Infection prevention and control reduces infection risk regardless of resistance profile — but the most valuable cases averted are those that would have been resistant to available antibiotics.
- If the infection prevention and control bundle averts 15,000 deaths annually, and ~20–30% of neonatal sepsis deaths in these settings are AMR-attributable, then ~4,500–6,000 deaths averted are AMR deaths.
Who could we fund to do this? ICAN (Infection Control Africa Network), a Pan-African infection prevention and control network working in hospitals, which has a strong implementation history.
Prescribing
Ensuring doctors are able to prescribe accurately (e.g. scaling access to diagnostics), and that they prescribe in a calibrated way (e.g. guidance and training to reduce over-, under-, or mis-prescription of antibiotics, or implementing “delayed prescribing” policies so that patients only receive antibiotics if their symptoms persist).
There are two general categories here: identifying when to prescribe antibiotics, and identifying the right antibiotics to prescribe.
Identifying when to prescribe antibiotics
The value of improving diagnosis for bacterial infections depends on the health benefits of averting an unnecessary antibiotic prescription. These come in two forms:
- Health benefits to an individual from avoiding an unnecessary antibiotic prescription
- Benefits from reducing aggregate AMR burden, by decreasing selective pressure on resistant pathogens
There are direct health benefits to the individual from avoiding antibiotics, including:
- Reduced risk of adverse drug reactions, which occur in ~5–25% of antibiotic courses, depending on the setting and antibiotic used. These reactions are occasionally severe.
- Avoidance of gut microbiome disruption, which may have long-term but hard-to-quantify consequences.
- Lower risk of future treatment failure due to reduced selection for individual-level resistance. (If you take antibiotics and a few bacteria are resistant enough to survive, they will multiply in your body, weakening further courses of the same antibiotic.)
However, in most evaluations, the per-person health gains from avoiding a single unnecessary course are modeled as negligible, especially in outpatient primary care contexts. This component is unlikely to meaningfully drive an intervention’s SROI.
Benefits from reducing aggregate AMR burden are the primary potential source of value from interventions to reduce unnecessary prescribing. The benefit comes from reducing selection pressure, and thereby reducing AMR-related health burden at a population level. Several studies attempt to estimate DALYs averted per course of antibiotics avoided, though this will vary based on the specific antibiotic and the location:
- Atkins et al. (2021, UK): Found that reducing 10.9 prescriptions per 10,000 person-years led to 128 DALYs averted annually, or ~0.0117 DALYs averted per antibiotic course avoided.
- Dickinson et al. (2021): Modeled CRP testing and estimated 0.0087–0.0115 DALYs averted per antibiotic course avoided, depending on whether patient and carer costs were included alongside health sector costs.
- Sense check: Dividing global estimates of AMR DALYs (~47 million) by estimated defined daily doses (~49.3 billion), yields ~0.0009 DALYs per course avoided. The implication of the sense check is that 0.01 is probably an upper bound (I doubt it is even that big), and even then this pencils poorly.
If we assume that avoiding an unnecessary antibiotic prescription is associated with roughly 0.01 DALYs in benefits, we would value it at $CG 1000, and we would be willing to pay ~$0.48 to achieve it (given our bar of 2100x). This likely means that interventions focused on reducing unnecessary antibiotic prescriptions are unlikely to be cost effective as a whole.
Realistically, this is an extremely high bar for interventions in this space. For example, diagnostics at a floor can cost $0.50, but they will only avert antibiotic use when they detect that an infection is viral rather than bacterial, and the diagnosis is followed. Adherence rates vary widely; one Vietnam trial, Do et al. (2016), showed ~64% adherence to negative CRP results.
This leads me to a bearish outlook on interventions designed primarily to reduce antibiotic prescribing through diagnostics or nudges — especially when antibiotics themselves are cheap, and downstream AMR effects are diffuse and delayed.
Identifying the right antibiotics to prescribe
An alternative use of diagnostics is not to identify cases where antibiotics are required (i.e. distinguish between bacterial and viral infections), but rather to determine whether a particular case is resistant to first-line antibiotics and therefore prescribe the right drug sooner.
However, based on a conversation with Chris Somerville and Heather Youngs, this type of diagnostic is very expensive at the moment (thousands of dollars, orders of magnitude higher than it would need to be to be feasible in LMIC settings). In addition, they believe it is relatively scientifically intractable to significantly reduce the cost of this type of diagnostic at the moment.
Below are some interventions in this general space:

Intervention: C-reactive protein (CRP) point-of-care testing
CRP POC testing is a cheap, fast, low-skill diagnostic that can be used to distinguish between bacterial and viral respiratory infections. High CRP suggests bacterial illness, supporting appropriate antibiotic use. These tests are especially relevant in LMICs, as an improvement on the most common approach to febrile respiratory illness (empirical treatment without any testing). CRP is inexpensive, rapid, and helps providers avoid many common prescription errors; it is arguably the most promising existing diagnostic for outpatient antibiotic stewardship.
However, based on a BOTEC using trial data from Vietnam and modeled AMR costs, CRP testing has a preliminary estimated SROI of ~240x, even under optimistic assumptions. See here for the full BOTEC.
- I modeled a potential intervention to scale C-reactive protein (CRP) point-of-care tests to reduce unnecessary antibiotic prescriptions in low-income country settings.
- I assumed CRP tests would be used on 2 million patients annually, focusing on those with suspected respiratory infections in primary care settings.
- I used data from Isaeva et al. (2025) and Do et al. (2016), which suggest that CRP testing reduces antibiotic prescriptions by approximately 24% in these contexts.
- Applying this reduction implies 480,000 antibiotic courses could be averted per year.
- To estimate health impact, I pulled from the best available DALY-per-course estimates: Atkins et al. (2021) (UK) and Dickinson et al. (2021) (Afghanistan), which suggest benefits in the range of 0.0087–0.0117 DALYs per course avoided.
- I used a round midrange estimate of 0.01 DALYs per course avoided. That implies 4,800 total DALYs averted annually (480,000 × 0.01), implying $480 million in CG value per year.
- I assume CRP tests would cost around $1.00 per test based on an OECD estimate of the cost of implementing CRP tests in LMIC settings, yielding a total annual cost of $2 million.
- Dividing CG value by cost, this gives an SROI of 240x — well below Coefficient’s bar of 2100x, even under relatively optimistic assumptions.
This illustrative BOTEC demonstrates the mechanics described above that make me bearish on efforts aimed at reducing prescriptions.
Quality and access
Quality and access interventions aim to ensure that people in LMICs receive antibiotics that are both available and effective. That is, patients can access the right drug, at the right time, in the right form — and that drug is not substandard or counterfeit. These interventions target a critical but under-resourced bottleneck in the AMR response: even when effective antibiotics exist, they often fail to reach patients in time, especially as resistant infections become more common.
This makes the quality and access space conceptually attractive. Unlike prescribing or stewardship — which typically act indirectly by slowing resistance or averting misuse — access-focused interventions directly reduce deaths from resistant infections. In many LMICs, AMR is not just a long-run threat of future superbugs; it is an ongoing crisis of avoidable mortality from already-resistant pathogens and broken delivery systems.
The key cruxes for whether interventions in this category are promising include:
- How often are resistant infections actually treatable with second-line or reserve antibiotics if they reach patients?
- How wide is the current access gap in practice — and what are realistic delivery rates?
- What is the cost per patient reached, especially in fragile or rural systems?
The BOTEC below, for second-line antibiotic provision for neonatal sepsis in sub-Saharan Africa, indicates that at least some access-focused interventions can meet a high cost-effectiveness bar. It estimates an approximate SROI of ~3,570x, and directly targets resistant infections responsible for tens of thousands of newborn deaths each year. This suggests that expanding access to proven drugs (rather than waiting for new ones) may be one of the more impactful paths for reducing AMR mortality in the near term. If the implementation challenges are tractable, this area likely warrants high prioritization within an AMR portfolio.
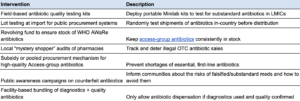
Intervention: Second-line antibiotic provision for neonatal sepsis in SSA
Neonatal sepsis is one of the leading causes of newborn death globally, and sub-Saharan Africa (SSA) bears a disproportionate share of that burden. Many cases are treatable with antibiotics — but the growing prevalence of resistance to first-line drugs (e.g. ampicillin and gentamicin) is making these standard regimens less effective. In cases where first-line treatment fails, access to second- or third-line antibiotics (such as cefotaxime, ceftriaxone, or carbapenems) can be life-saving.
However, in many parts of SSA, these broader-spectrum antibiotics are unavailable, unaffordable, or improperly administered. This creates a gap where newborns with resistant infections die, not because the drugs don’t exist — but because they aren’t reaching the patient in time. Ensuring wider access to second- and third-line antibiotics for neonatal sepsis is therefore a direct, high-impact way to reduce mortality from antimicrobial resistance in newborns.
Based on a BOTEC, providing second-line antibiotics for neonatal sepsis in sub-Saharan Africa may have an SROI of ~3,570x. See here for the full BOTEC.
- A figure of 40 million live births per year in sub-Saharan Africa and an incidence rate of neonatal sepsis of 2.8%, from Sands et. al (2022) implies ~1,129,600 neonatal sepsis cases annually in Sub-Saharan Africa.
- I assume ~50% of these cases are resistant to first-line antibiotics — drawn from Sherif et. al (2023) and GARDP, which report rates ranging from 40–85% of resistance in common neonatal sepsis pathogens in LMIC hospitals.
- I use an estimated ~25% mortality rate for resistant sepsis without access to second-line treatment and a ~15% mortality rate with access to appropriate second-line antibiotics based on Fleischmann et. al (2021), GARDP. This implies a 40% mortality reduction.
- I assume 70% of resistant cases would be reached with treatment, which implies that a total of ~790,720 resistant cases would be treated annually.
- Given the parameters above, this equates to 39,536 lives saved per year — or ~2 million DALYs averted. This intervention would therefore have an $CG value of ~$201.6 billion annually.
- I assume a cost per treatment of $100, based on estimates in Lashari et. al (2022) of ~$45 and scaling up since academic cost estimates tend to be structurally too low. This would imply a total cost of $56.5 million for all cases treated.
- This results in an SROI of 3,570x.
Who could we fund to do this? CHAI has worked on pediatric pneumonia drug access, and could likely extend that work to neonatal sepsis antibiotics (e.g. amikacin, meropenem).
Containment
Containment interventions aim to reduce the environmental spread of antimicrobial resistance by limiting the release of antibiotics and resistant bacteria into the environment — especially via pharmaceutical manufacturing waste, hospital effluent, and community sewage. These pathways can act as concentrated breeding grounds for resistance: antibiotics enter the environment at low doses, in the presence of diverse microbes and resistance genes, creating ideal conditions for the evolution and spread of resistant strains.
Antibiotic pollution is particularly concerning in LMICs, where gaps in both infrastructure and environmental regulation allow for higher levels of pharmaceutical pollution. Wilkinson et al. (2022) sampled 1,052 sites across 104 countries and found that four of the five worst-off rivers in terms of antibiotic contamination were in Africa.
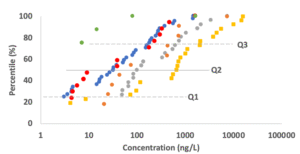
Concentrations of antibiotics in these rivers often far exceeded thresholds associated with both resistance selection and ecological harm. One study, Larsson et al. (2007), found ciprofloxacin concentrations near Indian pharmaceutical plants exceeding 30,000 ng/L — orders of magnitude above resistance-selection levels.
Environmental antibiotic levels are shaped by four factors: total use, waste disposal practices, treatment systems, and dilution capacity. In sub-Saharan Africa, the middle two factors — disposal and treatment — are the weakest links. Many rivers receive untreated sewage from hospitals, informal settlements, or entire urban populations. Fewer than 10% of wastewater streams are treated at all in several SSA countries. Even in major cities, discharge of raw or partially treated waste into the environment is common.
In contrast, in high-income countries (HICs), although antibiotic use is higher overall, most effluent is processed through wastewater treatment plants, which break down many compounds before they enter waterways. As a result, while we can’t say whether HICs or LICs have higher average antibiotic excretion volumes, the tails — the worst-case hotspots — are clearly worse in many LMICs. The available data is too skewed to allow meaningful comparison of global means, but the peaks in SSA and South Asia are indisputably extreme.
This presents a potentially serious risk. As studies like Hendriksen et al. (2019) and Wang et al. (2022) have shown, environmental antibiotic contamination is strongly associated with the spread of resistance genes in surface waters, sediments, and even drinking water sources. While there is limited evidence on the attribution of environmental contamination to human health burden, these pathways are biologically plausible and especially concerning in LMICs with poor sanitation and untreated effluent.
Whether containment is a promising area for AMR interventions depends on a few major uncertainties:
- What fraction of the AMR disease burden is caused by environmental transmission pathways? If the dominant source of resistant infections is patient-to-patient transmission (e.g. in hospitals), environmental containment may have limited impact. But if resistant bacteria from water, food, or soil are a substantial driver of human disease, containment could be crucial.
- How cost-effectively can containment interventions disrupt those transmission pathways? Can simple hospital infection prevention and control upgrades or low-cost wastewater treatment meaningfully reduce the spread of resistance? Or would meaningful reductions require expensive infrastructure and hard-to-enforce regulation?
- How measurable and attributable are the benefits? Because environmental AMR exposure is diffuse and long-term, many containment interventions suffer from weak evidence chains — making it difficult to directly estimate DALYs averted.
Containment currently appears unattractive from a cost-effectiveness standpoint. Mechanically, the challenge is that many containment interventions — such as wastewater treatment or industrial effluent controls — are capital-intensive, while the health benefits are diffuse, indirect, and difficult to attribute. Despite strong theoretical pathways linking environmental AMR contamination to human disease, the evidence base is too thin to support large DALY gains, and even optimistic BOTECs fall far below Coefficient’s bar.
There are two main ways this could change:
- First, if we’re able to rigorously quantify tail risk burdens from hotspot contamination — for example, identifying clusters where extreme environmental exposure causes direct outbreaks or widespread resistance amplification — and those risks are large enough to justify investment on expected value grounds, containment could become more compelling.
- Second, if future studies provide stronger empirical links between environmental AMR interventions and reductions in clinical infection or mortality, and those effects are larger than currently believed, it could shift the calculus.
Until then, containment likely remains a low-priority area for AMR-focused funding.
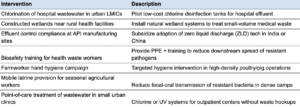
Intervention: Hospital wastewater chlorination
Hospitals are among the densest sources of antibiotic use in LMICs, and their untreated effluent often carries high loads of both antibiotics and resistant pathogens into surrounding waterways. This is especially true in urban areas where sewage systems are overburdened or nonexistent.
Chlorination tanks installed at hospital discharge points represent a low-tech, low-cost intervention to reduce microbial and chemical pollution — and, by extension, may help slow the environmental transmission of antimicrobial resistance. The potential public health value lies in interrupting resistance gene amplification and transmission via drinking water, agricultural use, and human contact.
I model a potential intervention to install low-cost chlorination systems at hospitals in high-burden LMIC cities to treat wastewater before release, aiming to reduce environmental AMR transmission, and find a preliminary SROI of ~825x. See here for the full BOTEC.
- I assume each hospital serves a catchment population of 100,000, reflecting dense, low-income urban areas where hospitals are embedded in residential zones and wastewater exposure is common.
- To estimate the total health burden, I use 30,000 DALYs / 100,000 people as a rough average for low-income countries, based on Global Burden of Disease 2019 estimates.
- I then attribute 5.5% of these DALYs to unsafe water, sanitation, and hygiene (WASH) risks, drawing on IHME’s 2019 WASH risk factor breakdowns.
- This implies ~1,650 WASH-related DALYs per year in each hospital’s catchment area.
- I then assume 5% of that WASH-related burden is attributable specifically to hospital wastewater, based on plausible but conservative attribution levels from Singer et al. (2016) and Berendes et al. (2020) and in line with the ~20% estimate used in AMR-specific models (e.g. Lamba et al. (2022)). This yields ~82.5 DALYs averted per hospital per year if wastewater is fully chlorinated.
- This translates into an estimated $8.25 million in CG benefit per hospital.
- I use a $10,000 per hospital cost estimate, based on WHO procurement data for low-cost chlorination systems, inclusive of installation and basic maintenance.
- This yields an SROI of ~825x, meaningfully below the bar.
I triangulated by estimating an AMR-specific impact and found it to be approximately ~189x, implying that ~22% of this impact comes from averting AMR-related DALYs.
- I assume each hospital has a catchment population of 100,000, reflecting the dense, urban settings where hospitals and nearby residents are often in close proximity to untreated effluent.
- I use an estimate of 18.5 AMR deaths per 100,000 population, based on the 2024 Lancet study on the global burden of bacterial AMR.
- That implies 18.5 AMR deaths per year in the catchment population per hospital.
- I assumed 10% of AMR deaths are attributable to environmental exposure, which is a rough estimate based on published reviews, including Singer et al. (2016) and Berendes et al. (2020). This implies 1.85 deaths annually from environmental AMR exposure per hospital catchment.
- I then estimate 20% of this environmental exposure comes from hospital wastewater, based on estimates in Lamba et al. (2022) and other LMIC urban wastewater tracking studies.
- That implies 0.37 AMR deaths per year or 18.87 DALYs attributable to hospital wastewater exposure. This yields a CG value of ~$1.89 million per hospital per year.
- I use a cost of $10,000 per hospital for chlorination infrastructure, based on WHO procurement estimates and low-tech chlorination system costs.
- This yields an SROI of 189x — far below the 2100x bar, even under optimistic assumptions.
Who could we fund to do this? icddr,b (Bangladesh) has deep technical expertise in WASH + AMR, especially in healthcare facilities.
The primary constraint is that the benefits to human health — especially AMR-related DALY burden — are difficult to attribute and small in magnitude. Even if hospital effluent contributes meaningfully to resistance in the environment, few studies offer direct links to clinical outcomes. If we were to get much stronger evidence on the link between environmental exposure and illness — or credible models to quantify it — that could significantly shift this estimate.
As it stands, this analysis supports the broader conclusion that containment interventions are structurally unattractive from a cost-effectiveness standpoint: they typically require infrastructure upgrades, and the health benefits — particularly AMR-related ones — are diffuse, indirect, and hard to measure.
Surveillance and governance
Most LMICs lack reliable data on how resistance patterns are shifting, which pathogens are driving disease, and where interventions are most needed. This information vacuum makes it difficult to guide prescribing, detect outbreaks, allocate resources, or evaluate policy effectiveness. As a result, antibiotics are often misused, outbreaks go uncontained, and resistant infections spread unchecked. At a higher level, the lack of granular data in LMICs makes it difficult for both philanthropic and public funders to allocate resources effectively.
While most HICs submit AMR data to the WHO’s Global Antimicrobial Resistance and Use Surveillance System (GLASS), fewer than half of LMICs do. Even where labs exist, data often comes from urban tertiary hospitals and is not integrated into routine decision-making. Many LMICs still lack basic systems for infection reporting, outbreak detection, or the enforcement of antibiotic regulations.
This category includes a range of interventions that aim to close these gaps:
- Surveillance systems: Expanding AMR lab networks, rolling out real-time resistance surveillance tools, or deploying genomic sequencing to detect early resistance clusters or novel strains.
- Data-driven response mechanisms: Setting up protocols for rapid action when resistance patterns change (e.g. switching first-line drugs, stockpiling second-line antibiotics).
- Outbreak containment: Triggering targeted responses in high-risk facilities (e.g. neonatal intensive care units) when resistance clusters emerge; using data to isolate, decolonize, and break transmission chains.
- Policy and regulatory reform: Advocating for evidence-based procurement practices, prescription monitoring, or national action plans that are grounded in surveillance data.
There are a few key cruxes that determine how attractive this area is for AMR-focused funding:
- Would better data actually change decisions? In principle, yes — but only if data is timely, actionable, and linked to mechanisms for response. Historical case studies show that early detection of resistance (e.g. rising carbapenem resistance) has enabled better treatment protocols and stockpiling decisions. But these cases are rare, and systematic evidence is thin. Surveillance alone doesn’t help unless it informs clinical or policy change.
- Are the benefits attributable and DALY-linked? Compared to other categories (like prescribing or containment), the causal chain here is shorter when surveillance is tied to concrete actions like outbreak control or switching antibiotics. For example, a hospital that uses surveillance to detect and contain a carbapenem-resistant Klebsiella outbreak can prevent dozens of deaths. But general improvements in data quality or national dashboards are harder to link to measurable DALY gains.
- Are costs manageable? Yes. While national systems can be expensive, targeted surveillance in high-burden hospitals or for high-priority pathogens is relatively cheap. The best interventions in this space are often narrow and low-cost, with high upside.
Surveillance and governance interventions are moderately attractive for AMR-focused funding. The strongest plays are targeted: bundling data systems with rapid infection prevention and control response in hospitals, or funding diagnostics and genomic tools for early resistance detection. These can produce short, attributable causal chains. In contrast, diffuse governance reforms or national dashboards — while important — are less likely to meet our cost-effectiveness bar without a clear link to behavior change or mortality reduction. Overall, this is a promising area when tightly scoped and operationally grounded in decision-making. Loose or purely informational efforts are less likely to pay off.
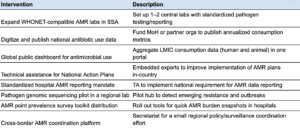
Intervention: Surveillance and infection control in ICU settings
Hospitals in LMICs face high rates of healthcare-associated infections (HAIs), many of which are caused by resistant pathogens and lead to preventable deaths. This burden is especially concentrated in intensive care units and surgical wards, where infection risk is highest and empirical antibiotic use is common.
By pairing routine surveillance of infection trends with basic infection prevention and control (IPC) practices — such as hand hygiene, PPE, and stewardship protocols — facilities can rapidly detect outbreaks, adjust antibiotic use, and reduce the overall incidence of HAIs. Unlike broader “containment” or “prescribing” interventions, which face long and uncertain pathways to DALY impact, hospital-based surveillance and infection prevention and control bundles offer a short, measurable causal chain: fewer infections, fewer resistant cases, and fewer deaths.
I modeled a potential intervention to expand surveillance and infection control programs for healthcare-associated infections in LMIC hospitals, drawing on bundled approaches that combine real-time infection monitoring with basic infection prevention and control upgrades (e.g. hygiene, PPE, antibiotic stewardship) and estimate an SROI of ~1986x. See here for the full BOTEC.
- I (illustratively) assume this intervention is rolled out across 1,000 tertiary hospitals in LMICs, covering a total of 10 million annual inpatient admissions (10,000 per hospital).
- Based on a meta-analysis by Schreiber et al. (2018), enhanced HAI surveillance combined with infection control can reduce HAI incidence by 35–55%. I use an estimate of ~40%.
- I use an estimated baseline incidence of 15% HAI for inpatients in LMIC hospitals, based on the 2022 WHO report. That implies 1.5 million HAIs annually in this population, with 450,000 averted due to the intervention.
- I estimate a 15% baseline additional mortality rate for patients who get HAIs (i.e. 15% of those patients end up dying when they wouldn’t have without the infection). This implies 67,500 deaths averted through averting 450,000 infections.
- The International Nosocomial Infection Control Consortium (INICC) reported that in LMIC ICUs, patients without HAIs had a mortality rate of ~17%, while patients with one HAI had mortality rates ranging from 30–48%.
- Taking the average of these two values would imply a marginal increase of 20% in mortality rate attributable to HAIs. I use 15% as a more conservative estimate.
- Using 32 DALYs per death, this results in 2.16 million DALYs averted. At $100,000 per DALY, the total CG value of the intervention is $216 billion.
- I assume a cost of $161,000 per hospital per year from Stone et. al (2010), though I have significant uncertainty around this figure. This would cover a simple monitoring system, staff training, and basic infection prevention and control upgrades. This yields a total cost of $50 million annually across 1,000 hospitals.
- This results in an SROI of ~1986x.
Who could we fund to do this? The CDC Foundation, which partners with LMIC health ministries on hospital surveillance systems.
Diagnostic and drug development
AMR is driven by overuse, underperformance, and lack of existing antibiotics. Diagnostic and drug development interventions aim to tackle this from both ends: enabling more accurate prescribing and expanding the antibiotic toolbox. This includes both push and pull mechanisms:

The portion of the AMR burden this category aims to reduce includes:
- Drug-resistant infections that cannot be treated with available antibiotics (e.g. extensively drug-resistant Klebsiella or Acinetobacter).
- Infections misdiagnosed or mistreated due to lack of rapid, accurate pathogen data, leading to incorrect prescriptions or harmful delays.
The majority of diagnostics are likely to have low SROI for structural reasons. Despite their intuitive appeal, most diagnostics perform poorly in cost-effectiveness terms for AMR. This is for two main reasons:
- Most of the benefit from accurate diagnosis accrues to the individual patient, not to population-level resistance. Avoiding one unnecessary antibiotic course has very little measurable impact on the AMR burden. That makes most diagnostics similar in structure to prescribing behavioral interventions, which tends not to be very cost-effective.
- Where diagnostics do avert deaths (e.g. via faster sepsis treatment), the benefit is real but not necessarily AMR-specific. These DALYs are valid but may be outside the scope of AMR-targeted funding, and many funders already support general diagnostic scale-up.
The primary potential exception is diagnostics that directly enable effective treatment of resistant infections — particularly when second-line drugs are available but would not be used unless the patient was diagnosed. For example, in neonatal sepsis, where Klebsiella and Acinetobacter are often resistant to first-line antibiotics, a rapid diagnostic that detects these pathogens or resistance markers could trigger life-saving therapy. In these cases, the diagnostic is not just reducing unnecessary use, but enabling timely treatment that would not otherwise occur — making the DALY impact both measurable and AMR-specific.
However, based on my conversation with Chris Somerville and Heather Youngs, diagnostics that identify treatment resistance are currently extremely expensive (at thousands of dollars per test, orders of magnitude more than would be feasible for adoption in LMICs), and appreciably reducing the cost is likely scientifically intractable at the moment.
Drug development is more promising, but only for certain elements of the burden. A natural objection is that access is the real problem in LICs — not a lack of drugs. In many cases, that’s true: resistant infections are still treatable with existing antibiotics like carbapenems, and the barrier is health system failure. But this isn’t always the case. Some infections — like neonatal Klebsiella — are already resistant to everything available or safe in LICs.
Drug development still matters for a subset of the AMR burden where:
- Existing antibiotics are no longer effective (e.g. extensively drug-resistant Klebsiella in neonates)
- Available treatments are too toxic for vulnerable populations (e.g. neonates, pregnant women)
- Pathogens are spreading in settings where transmission is high
The clearest example is neonatal sepsis. According to the 2022 Lancet study, drug-resistant Klebsiella pneumoniae is responsible for ~80,000 neonatal deaths annually, many in LICs. In these cases, access to second-line drugs isn’t enough—because those second-line drugs don’t work, or aren’t approved for neonates.
A study by Folgori et al. (2017) found that only 1 of 40 antibiotics in the pipeline was being developed for use in neonates. A more recent pipeline review by the WHO (2023) finds a similar trend: almost all new antibiotics are designed for adult use in high-income countries, with little attention to pediatric populations or pathogens found largely within LMICs.
This suggests drug development can be cost-effective when:
- It targets high-burden, under-addressed pathogens in LMICs.
- It focuses on reformulations or trials of drugs that already exist for adults but haven’t been validated for neonates or children.
- It is paired with mechanisms to ensure affordable LMIC rollout.
In these cases, the intervention addresses the upstream constraint: the absence of a usable, safe, and effective drug.
Based on my conversation with Chris and Heather, they generally believe antibiotic development to be less cost-effective than the work they currently fund — and likely below the Coefficient bar — based in part on a lack of neglectedness, and on the cyclical nature of antibiotics (i.e. each one produced will only be effective for a limited time). However, there may be room for specific interventions to be cost-effective if they fill a niche neglected by CARB-X and GARDP.
Finally, note that there are some experimental alternatives to antibiotics, such as bacteriophages and CRISPR. However, my conversation with Chris and Heather confirmed these are unlikely to be attractive areas for investment: they have the same selection pressures as antibiotics and are — all else equal — less effective. They are mostly in use because they have (to date) been used less than antibiotics and bacteria are less likely to resist them so far. But any scale-up of the treatment would change that.
The AMR drug development ecosystem includes a mix of philanthropic, multilateral, and public-sector actors. While some (like GARDP and Wellcome) are explicitly focused on LMICs, others prioritize global pipelines or HIC markets. Each group tends to concentrate on specific stages of development.
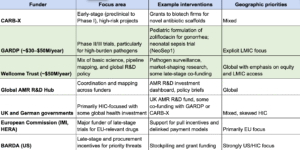
Based on this initial mapping, my best guesses for the gaps in funding are:
- Late-stage trials for LMIC-priority infections (especially neonatal sepsis)
- GARDP is one of the only actors here, and they can’t fund more than a few candidates at a time.
- Many high-burden LMIC pathogens (e.g. Klebsiella pneumoniae, Acinetobacter) lack late-stage candidates.
- Rollout, access, and manufacturing in LICs
- No major funder is focused on bridging the gap between regulatory approval and real-world access in LICs.
- Little support is available for local/regional manufacturing capacity, pediatric or heat-stable formulations, or health system integration and procurement.
- Market-shaping and pull incentives for LMIC use
- Most pull incentives (e.g. BARDA subscriptions, EU delinkage pilots) are HIC-focused.
- No equivalent exists to guarantee uptake or procurement in LICs — meaning that even effective drugs may go unused.
- Stewardship-compatible drug development
- Narrow-spectrum or “pathogen-targeted” antibiotics are disincentivized by the current market model.
- These drugs often won’t be profitable and receive little attention from commercial actors or early-stage funders.
- Reformulations and older drug optimization
- There is little funding for “non-innovative” but potentially high-impact work: for example, retesting older, off-patent antibiotics in new combinations, dosage forms, or delivery mechanisms.
- These projects fall through the cracks between R&D and implementation funding.
I’m not highly confident in this list — I’d speak to someone at GARDP if I wanted to refine it — but I hope it can still help to guide identification of promising interventions in this category.
Overall, diagnostic and drug development interventions have highly variable SROI. Diagnostics are structurally weak on SROI and unlikely to exceed Coefficient’s bar unless tightly tied to AMR-specific DALYs. Drug development is more promising — while some organizations (notably GARDP and Wellcome) are doing significant work in the space, the global pipeline remains heavily skewed toward HIC priorities, early-stage novelty, and marketable candidates. As a result, the highest-return opportunities may lie not in starting new pipelines, but in pushing known, LMIC-relevant candidates over the finish line — especially for high-burden, under-addressed conditions like neonatal sepsis.

Intervention: Phase II/III trial of a narrow-spectrum antibiotic for neonatal sepsis
Neonatal sepsis is one of the deadliest and most drug-resistant infections in low- and middle-income countries, causing an estimated ~20 million DALYs annually and approximately 370,000 deaths globally, many of which are resistant to first-line antibiotics (GRAM 2022). The most common pathogens — Klebsiella pneumoniae, Acinetobacter baumannii, and E. coli — often show 40–85% resistance to recommended empiric antibiotics in LMIC hospitals (Sherif et al. (2023); GARDP surveillance reports).
However, the antibiotic pipeline structurally includes less research into treatments for neonates due to heightened safety concerns, and potentially profit disincentives. A review by Folgori et al. (2017) found that only 1 of 40 antibiotics in development was being studied for neonatal use. WHO’s 2023 clinical pipeline report shows this hasn’t changed: nearly all new drugs are being developed for adult use in high-income countries, with no late-stage candidates targeting neonatal sepsis in LMICs beyond one GARDP-led trial (NeoSep1). GARDP’s funding (~$30–50 million/year) allows it to support only a single Phase III trial at a time.
That leaves strong candidates untested. For example:
- Cefepime/taniborbactam (Venatorx) and cefiderocol (Shionogi) both have promising activity against carbapenem-resistant Klebsiella and Acinetobacter.
- Neither is approved for neonates, and no publicly funded trials are validating them for neonatal sepsis.
- Commercial firms are unlikely to pursue neonatal indications in LMICs due to high regulatory burden and low return.
The burden of neonatal sepsis is high not only because of infection, but also because of comorbidities like preterm birth (involved in 56% of neonatal sepsis deaths per CHAMPS) and perinatal asphyxia (16%). However, modeling by Rudd et. al suggests that the sepsis-attributable fraction remains ~80%, even after accounting for these risk factors.
Neonatal Klebsiella pneumoniae sepsis alone may account for ~5 million DALYs annually, given its role as the dominant pathogen in 27–46% of bacteremia cases (CHAMPS, BARNARDS, NeoObs studies). This suggests that developing and validating a narrow-spectrum drug targeting this and other major pathogens could yield high impact.
I estimated the impact of a potential intervention to fund a Phase II or III trial of a narrow-spectrum drug for neonatal sepsis, and find it may have an SROI of ~1,699x. See here for the full BOTEC.
- There are ~1.13 million neonatal sepsis cases annually in sub-Saharan Africa, based on ~40 million births per year and an incidence rate of 2.8% (Sands et al. (2022)).
- I assume ~50% of these are resistant to first-line antibiotics (range: 40–85%)
- I use an estimated ~25% mortality rate for resistant sepsis without access to second-line treatment and a ~15% mortality rate with access to appropriate second-line antibiotics based on Fleischmann, et. al (2021) and GARDP. This implies a 40% mortality reduction.
- Assuming 50% treatment coverage, ~790,720 babies would receive the new drug annually.
- That implies ~39,536 lives saved per year, or ~2 million DALYs averted, using 51 DALYs per neonatal death (Coefficient BOTEC Guide).
- At $100,000 per DALY, the annual Coefficient value of the intervention is $201.6 billion.
- I assume a total cost of $100 million to cover the trial and scale-up in LMIC settings, based on existing Phase III trial budgets and rollout estimates. I also assume a 15% chance of failure in Phase III.
- This implies an SROI of ~1,214x.
Existing neonatal drug development has systematically underprioritized LMIC neonatal sepsis despite Klebsiella and Acinetobacter accounting for a substantial portion of neonatal deaths. GARDP is only testing one candidate (fosfomycin + amikacin), leaving others like cefepime/taniborbactam unvalidated in neonates. Funding a second trial could have significant additive value.
Who could we fund to do this? GARDP (Global Antibiotic R&D Partnership) to expand to another trial.
Animal agriculture
Animal agriculture is one of the largest global consumers of antibiotics. Roughly 70% of all antibiotics by weight are estimated to be used in livestock — largely for non-therapeutic purposes like growth promotion and infection prevention in high-density farming operations. In low- and middle-income countries (LMICs), where livestock production is expanding rapidly and regulatory systems are often weak, the use of medically important antibiotics in animals remains common.
There is significant variation in how often antibiotics are used in animal agriculture, potentially suggesting that targeting the countries with the highest use might yield outsized benefits. Asia, Oceania, and most of the Americas use a lot of antibiotics, while Europe and Africa tend to use less than 50 milligrams per kilogram.
This is mostly driven by affordability and access, differences in regulatory and industry norms, and the most popular types of livestock in a given country or region.
However, while the volume of use is large, the actual contribution of animal antibiotic use to human AMR disease burden is less clear. Many antibiotics used in animals (e.g. ionophores) are not used in human medicine. Even when human-relevant antibiotics are used, the degree to which resistant bacteria or resistance genes transfer to people — and result in clinical infections — remains poorly quantified. Most transmission chains from livestock to humans are thought to be indirect, via the environment, food handling, or occupational exposure, and likely account for only a small fraction of total resistant infections.
Studies like Smith et al. (2005) and the AMR Review (O’Neill, 2015) suggest that agricultural antibiotic use may contribute to a small minority of human AMR deaths — perhaps 5–10% — and that curbing animal use may only modestly reduce human AMR burden. No studies explicitly estimate that agricultural antibiotic use contributes to much more than 10% of human AMR deaths. I wouldn’t take this as conclusive, there is a lot of uncertainty around attribution, but I would say we don’t have good evidence that it is a large driver at this point.
This category includes several types of interventions:
- Reduce the need for antibiotics
-
-
- Vaccination programs: Broaden access to livestock vaccines to prevent common infections (e.g. Salmonella, Pasteurella).
- Improved animal health & husbandry: Enhance sanitation, ventilation, biosecurity, and feeding practices to reduce infection risk.
- Alternatives to antibiotics: Introduce prebiotics, probiotics, or other growth-promoting feed additives that don’t carry resistance risks.
-
- Regulate or limit antibiotic use
-
-
- Targeted bans or restrictions: Advocate for bans on the use of medically important antibiotics (e.g. colistin) for non-therapeutic purposes.
- Use guidelines and stewardship: Develop or enforce prescribing guidelines for veterinary antibiotic use, especially in poultry and pigs.
- Monitoring & reporting systems: Build systems to track antibiotic sales or usage on farms and identify hotspots of overuse.
-
- Incentivize reduced use of antibiotics
-
- Subsidies or payments for reduced-use production: Pay farmers to pilot antibiotic-free production models with external verification.
- Market-based certification and labeling: Create third-party certification schemes for antibiotic-free meat to reward compliant producers.
- Public procurement standards: Tie government meat procurement to antibiotic use standards, nudging large-scale suppliers.
Key cruxes that inform the attractiveness of interventions related to AMR in agriculture are:
- How much does animal antibiotic use contribute to human AMR burden? Likely only a small fraction. While plausible transmission routes exist, empirical evidence suggests that hospital and community antibiotic use in humans — not agriculture — is the dominant driver of resistant clinical infections.
- Is it feasible to change animal antibiotic use in LMICs? Very difficult. Many LMICs rely on intensive animal production for food security, and farms operate on thin margins. Government enforcement is often weak, industry lobbying is strong, and global demand for cheap meat continues to rise.
- Can we measure benefits and ensure compliance? No. Unlike with health facility interventions, it is very difficult to verify reductions in antibiotic use or link those reductions to DALYs averted. Monitoring is expensive and compliance is difficult to enforce, especially in informal or smallholder farming systems.
Animal agriculture is structurally similar to prescribing and containment: the goal is to reduce selection pressure and slow resistance emergence, rather than directly averting current deaths. As with those areas, the link between the intervention and DALYs averted is long, indirect, and poorly evidenced. But animal agriculture is likely even less tractable. The barriers to implementation are steep — including food security concerns, powerful industry actors, and very limited regulatory capacity. Interventions like bans, farm training, or vaccine promotion are unlikely to scale meaningfully in the near term.
Taken together, the empirical uncertainty, low tractability, and difficulty in attributing DALY impact make this category structurally and practically unattractive — even more so than prescribing or containment. Given its high structural and practical hurdles, weak empirical links to human disease burden, and similarity to other already-unattractive categories, our current view is that animal agriculture is a weak candidate for AMR-focused funding.
From a FAW perspective, the picture is uncertain. Martin Gould’s take is that, in the US, lower levels of antibiotic use don’t clearly lead to higher welfare, and he has heard that organic farms have worse cow welfare due to a lack of antibiotic usage. Lewis Bollard has a thread on what happened when usage went down in the broiler industry (ultimately bad things for chicken welfare). This might suggest that reducing antibiotic usage could be somewhat negative from an animal welfare point of view.
I believe the mechanical reasons for this are sufficiently demonstrated by the prescribing and containment BOTECs, so I do not BOTEC an animal agriculture intervention. However, below are some example interventions in this space.
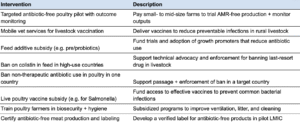
High value-of-information opportunities
This section addresses the question ‘What additional research would be most likely to change the conclusions in this investigation in ways that would directly alter our decisions?’
The biggest key uncertainty, that impacts nearly all estimates, is: ‘What actually causes the AMR burden?’ Specifically, what share of the burden is attributable to different drivers (e.g. environmental exposure, human antibiotic misuse, animal agriculture, transmission pathways), and what is the marginal impact of interventions targeting each?
Research questions that would help inform this work, and be relatively high value-of-information:
-
- Cross-cutting: What are the relative contributions of different drivers to the AMR disease burden in LMICs?
- What share is due to inappropriate antibiotic use in humans?
- What share is due to lack of access to effective antibiotics?
- What share is due to environmental exposure (e.g. water, soil, food)?
- What share is due to antibiotic use in animal agriculture?
- Prescribing: What is the DALY impact of averting a single unnecessary antibiotic course in LMIC outpatient settings?
- Quality and access: How much do second-line antibiotics reduce mortality in neonatal sepsis cases resistant to first-line drugs?
- Quality and access: What are the real-world access rates and delivery bottlenecks for second-line antibiotics for neonatal sepsis in SSA?
- Surveillance and governance: What decisions at the hospital or national level in LICs could be meaningfully improved by better AMR surveillance data?
- Diagnostics and drug development: How much can faster identification of first-line-resistant pathogens improve clinical outcomes in neonatal sepsis?
- Diagnostics and drug development: Which Phase II/III antibiotics for bacterial infections are stalled due to lack of funding — particularly for validation in e.g. neonates – and how promising are they?
- Cross-cutting: What are the relative contributions of different drivers to the AMR disease burden in LMICs?
Conclusions and next steps
This shallow investigation finds that antimicrobial resistance (AMR) is a high-burden global health issue, particularly in low- and middle-income countries (LMICs), with an estimated 32 million DALYs attributable to AMR annually, two-thirds to three-fourths of which fall in LMICs. Public and philanthropic spending on AMR is relatively low at ~$43/DALY globally, putting it on par with areas like tuberculosis, and ~$7–10.50/DALY in LMICs. A majority of the existing funding is concentrated in R&D, particularly for diagnostics and drugs, while access- and implementation-focused work in LMICs remains underfunded.
The most promising areas for philanthropic investment appear to be:
- Prevention, particularly when targeting resistant pathogens (e.g. chlorhexidine, typhoid conjugate vaccines, infection prevention and control bundles in neonatal intensive care units),
- Quality and access, particularly expanding second-line antibiotics for neonatal sepsis in sub-Saharan Africa,
- Select opportunities in surveillance and governance and drug development, when tightly scoped and tied to decision-making or late-stage trials.
To get to ‘yes’ on AMR as a priority cause area, we would want to:
- Confirm that multiple grant opportunities in prevention, access, surveillance, or drug development reliably clear the bar, both conceptually and in execution.
- Determine that these are not isolated opportunities, but represent an ongoing, scalable pipeline of impactful grants. We would need to believe we could spend ~$10 million/year above the bar on those areas.
- Understand what an overarching programmatic strategy would look like across the most appealing areas of AMR funding, and how this would fill a gap within the current ecosystem of funders (including why, e.g., Wellcome is not funding it).
- Resolve key uncertainties about the drivers of AMR burden and the attribution of impact from different intervention types.
The biggest cross-cutting uncertainty is the relative attribution of AMR burden — that is, how much of the health burden stems from different causes (e.g. inappropriate prescribing, environmental exposure, lack of access to second-line drugs). Other important uncertainties include:
- The real-world effect size and delivery feasibility of high-priority interventions (e.g. second-line antibiotic provision).
- The marginal value of averting one unnecessary antibiotic course, which underpins many prescribing-focused interventions.
- The decision-making value of better surveillance data in LMIC contexts.
- Which drug candidates are bottlenecked at late-stage trial or pediatric formulation and might be cost-effective to fund.
Suggested next steps:
- Make or explore small grants now in prevention, quality and access, or surveillance that appear comfortably above the bar.
- Pursue a few targeted high-value-of-information grants, such as funding research on the relative drivers of AMR burden, or understanding access bottlenecks for second-line antibiotics.
- If initial grants go well, do a medium-depth investigation into one or more of the most promising intervention areas, with the goal of assessing whether we could build a $10 million+/year program based on that area.
Economic impact estimate methodology and pressure-testing
How did they get these figures? In order to get these values, the authors combine updated human-health burden forecasts from the IHME with economic modeling across multiple channels. First, they calculate inpatient care costs by synthesizing cost-of-illness data from hundreds of studies, mapped to resistant cases in different countries and adjusted for local health spending. Second, they model several “resilience” pathways (including labor, tourism, and hospitality), drawing on both a literature review of comparable diseases and an expert elicitation of how people might change their behaviors if resistance worsened. Third, these estimates feed into a computable general equilibrium (CGE) model that captures ripple effects across global economies. Finally, the authors examine several scenarios — ranging from “business as usual” to ones involving widespread improvements in treatment, innovation, vaccines, and hygiene — and estimate both the incremental cost of these interventions and their resultant health and economic gains.
Should we trust the methodology? Although this approach is more granular than previous economic AMR studies (which are pretty limited), there are reasons it could be an over- or underestimate. The reliance on hospital cost data (especially in low-income contexts) risks undercounting community-level impacts, so the real health and economic burden might be higher. Conversely, the behavioral assumptions — particularly about how rapidly people and industries would adapt — could mean some losses might be smaller than modeled in the long run. The report acknowledges that there is substantial uncertainty, especially around country resilience scores and the exact pace of new antibiotic development.
Are the estimates reasonable based on other AMR estimates? The overall findings appear directionally consistent with earlier AMR estimates from the World Bank and the 2014 AMR Review, which also forecasted large global losses. An AMR Review (O’Neill, 2014/16) projected a $100 trillion cumulative GDP shortfall (roughly 3–4% of global GDP in 2050). Note that Anthony McDonnell, who worked on this paper, believes this is an overestimate. That said, that worst-case forecast is significantly larger than CGD’s “accelerated” scenario of a ~0.8% GDP hit in 2050. The World Bank’s (2017) “low” vs. “high” AMR scenarios showed 1.1–3.8% global GDP loss by mid‐century — again bigger than CGD’s ~0.8% in the “accelerated” case. The World Bank’s modeling approach made more severe assumptions for the high-resistance scenario, which plausibly inflates the top-end figure.

CGD’s new estimate (<1% of GDP in the worst scenario) is more conservative than those earlier estimates, though it is still large in absolute terms. The lower figure is likely more accurate; the CGD team integrated more up-to-date AMR burden data, and – for the projections – and ran more moderate “accelerated” parameters than O’Neill or the World Bank’s highest‐plausibility scenario.
Are the estimates reasonable based on triangulating with other disease areas? Other high-burden infectious diseases, such as HIV or malaria, have an estimated comparable economic threat in developing regions. For example, Bell, Devarajan, and Gersbach (2003) conclude that HIV could reduce the per-capita GDP growth rate by 1%–2% per year in some African states, compounding over a few decades. Though, of course, HIV/AIDS in 2025 is very different from the problem in 2003. Estimates from Gallup and Sachs (2001) suggested that malaria-endemic countries grew ~1.3% per year more slowly than non-endemic countries over a long horizon. The WHO Macroeconomics and Health Commission (2002) also suggested that in heavily malaria-affected regions, the disease could depress annual GDP growth by more than a full percentage point.
These are focused on the hardest-hit countries, while the AMR estimate is global. However, HIV and malaria are both relatively concentrated in burden. This means taking the GDP impact of malaria in the hardest-hit countries and comparing it to an estimate of the global AMR economic impact would be biased upward (i.e. the malaria burden would look higher even if its overall impact on the world were similar to AMR’s). As a result, given that HIV and malaria have similar estimated global DALY burdens to AMR (~40–60 million range), the fact that they have a similar if slightly higher estimated economic burden lends some credence to CGD’s estimates.
How does this impact our previous top-line figure for AMR importance? We think of the CG value of income effects as X*($50,000/i), where X is the total dollar loss, and ‘i’ is the annual income of people experiencing the loss. However, in this case identifying the right ‘i’ to use is tricky. Considering direct costs, high-income countries spend the most on healthcare per capita, and so are likely to experience more of the cost of AMR. However, LMICs experience a much higher health burden (DALYs and deaths) due to AMR, which is a countervailing factor. LMICs of course also have a higher absolute population, meaning the average will be weighted toward those countries. Understanding that this is an oversimplification, I’ll use the average global annual income of $23,380. In this case, the CG value would be $66 billion*($50,000/$23,380) = ~$141 billion. Adding direct economic costs (i.e. healthcare costs) to health effects would bring the total attributable burden of AMR in CG terms from $3.2–$3.34 trillion.